Evidence that intracellular stages of Leishmania major utilize amino sugars as a major carbon source
- PMID: 21203480
- PMCID: PMC3009595
- DOI: 10.1371/journal.ppat.1001245
Evidence that intracellular stages of Leishmania major utilize amino sugars as a major carbon source
Abstract
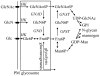
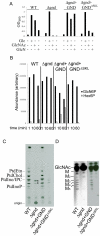
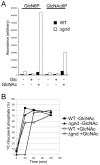
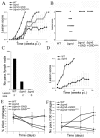
Intracellular parasites, such as Leishmania spp, must acquire suitable carbon sources from the host cell in order to replicate. Here we present evidence that intracellular amastigote stages of Leishmania exploit amino sugars in the phagolysosome of mammalian macrophages as a source of carbon and energy. L. major parasites are capable of using N-acetylglucosamine and glucosamine as primarily carbon sources and contain key enzymes required for conversion of these sugars to fructose-6-phosphate. The last step in this pathway is catalyzed by glucosamine-6-phosphate deaminase (GND), which was targeted to glycosomes via a canonical C-terminal targeting signal when expressed as a GFP fusion protein. Mutant parasites lacking GND were unable to grow in medium containing amino sugars as sole carbohydrate source and rapidly lost viability, concomitant with the hyper-accumulation of hexosamine-phosphates. Expression of native GND, but not a cytosolic form of GND, in Δgnd parasites restored hexosamine-dependent growth, indicating that toxicity is due to depletion of glycosomal pools of ATP. Non-lethal increases in hexosamine phosphate levels in both Δgnd and wild type parasites was associated with a defect in promastigote metacyclogenesis, suggesting that hexosamine phosphate levels may influence parasite differentiation. Promastigote and amastigote stages of the Δgnd mutant were unable to replicate within macrophages and were either completely cleared or exhibited reduced lesion development in highly susceptible Balb/c mice. Our results suggest that hexosamines are a major class of sugars in the macrophage phagolysosome and that catabolism of scavenged amino sugars is required to sustain essential metabolic pathways and prevent hexosamine toxicity.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures




References
-
- Swanson MS, Fernandez-Moreira E. A microbial strategy to multiply in macrophages: the pregnant pause. Traffic. 2002;3:170–177. - PubMed
-
- Schaible UE, Kaufmann SH. A nutritive view on the host-pathogen interplay. Trends Microbiol. 2005;13:373–380. - PubMed
-
- Appelberg R. Macrophage nutriprive antimicrobial mechanisms. J Leukoc Biol. 2006;79:1117–1128. - PubMed
-
- Naderer T, McConville MJ. The Leishmania-macrophage interaction: a metabolic perspective. Cell Microbiol. 2008;10:301–308. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

