Mitochondrial enzymes are protected from stress-induced aggregation by mitochondrial chaperones and the Pim1/LON protease
- PMID: 21209324
- PMCID: PMC3046053
- DOI: 10.1091/mbc.E10-08-0718
Mitochondrial enzymes are protected from stress-induced aggregation by mitochondrial chaperones and the Pim1/LON protease
Abstract
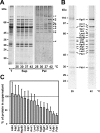
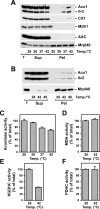
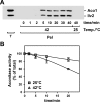
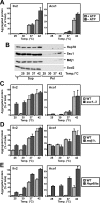
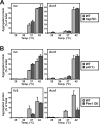
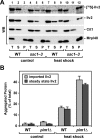
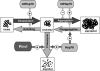
Proteins in a natural environment are constantly challenged by stress conditions, causing their destabilization, unfolding, and, ultimately, aggregation. Protein aggregation has been associated with a wide variety of pathological conditions, especially neurodegenerative disorders, stressing the importance of adequate cellular protein quality control measures to counteract aggregate formation. To secure protein homeostasis, mitochondria contain an elaborate protein quality control system, consisting of chaperones and ATP-dependent proteases. To determine the effects of protein aggregation on the functional integrity of mitochondria, we set out to identify aggregation-prone endogenous mitochondrial proteins. We could show that major metabolic pathways in mitochondria were affected by the aggregation of key enzyme components, which were largely inactivated after heat stress. Furthermore, treatment with elevated levels of reactive oxygen species strongly influenced the aggregation behavior, in particular in combination with elevated temperatures. Using specific chaperone mutant strains, we showed a protective effect of the mitochondrial Hsp70 and Hsp60 chaperone systems. Moreover, accumulation of aggregated polypeptides was strongly decreased by the AAA-protease Pim1/LON. We therefore propose that the proteolytic breakdown of aggregation-prone polypeptides represents a major protective strategy to prevent the in vivo formation of aggregates in mitochondria.
Figures

References
-
- Balaban RS, Nemoto S, Finkel T. Mitochondria, oxidants, and aging. Cell. 2005;120:483–495. - PubMed
-
- Bateman JM, Iacovino M, Perlman PS, Butow RA. Mitochondrial DNA instability mutants of the bifunctional protein Ilv5p have altered organization in mitochondria and are targeted for degradation by Hsp78 and the Pim1p protease. J Biol Chem. 2002;277:47946–47953. - PubMed
-
- Bender T, Leidhold C, Ruppert T, Franken S, Voos W. The role of protein quality control in mitochondrial protein homeostasis under oxidative stress. Proteomics. 2010;10:1426–1443. - PubMed
-
- Bota DA, Davies KJ. Lon protease preferentially degrades oxidized mitochondrial aconitase by an ATP-stimulated mechanism. Nat Cell Biol. 2002;4:674–680. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

