Partitioning of 2,6-Bis(1H-Benzimidazol-2-yl)pyridine fluorophore into a phospholipid bilayer: complementary use of fluorescence quenching studies and molecular dynamics simulations
- PMID: 21211898
- PMCID: PMC4167733
- DOI: 10.1016/j.bpc.2010.12.001
Partitioning of 2,6-Bis(1H-Benzimidazol-2-yl)pyridine fluorophore into a phospholipid bilayer: complementary use of fluorescence quenching studies and molecular dynamics simulations
Abstract
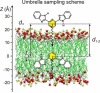
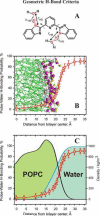
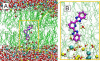
Successful use of fluorescence sensing in elucidating the biophysical properties of lipid membranes requires knowledge of the distribution and location of an emitting molecule in the bilayer. We report here that 2,6-bis(1H-benzimidazol-2-yl)pyridine (BBP), which is almost non-fluorescent in aqueous solutions, reveals a strong emission enhancement in a hydrophobic environment of a phospholipid bilayer, making it interesting for fluorescence probing of water content in a lipid membrane. Comparing the fluorescence behavior of BBP in a wide variety of solvents with those in phospholipid vesicles, we suggest that the hydrogen bonding interactions between a BBP fluorophore and water molecules play a crucial role in the observed "light switch effect". Therefore, the loss of water-induced fluorescence quenching inside a membrane are thought to be due to deep penetration of BBP into the hydrophobic, water-free region of a bilayer. Characterized by strong quenching by transition metal ions in solution, BBP also demonstrated significant shielding from the action of the quencher in the presence of phospholipid vesicles. We used the increase in fluorescence intensity, measured upon titration of probe molecules with lipid vesicles, to estimate the partition constant and the Gibbs free energy (ΔG) of transfer of BBP from aqueous buffer into a membrane. Partitioning BBP revealed strongly favorable ΔG, which depends only slightly on the lipid composition of a bilayer, varying in a range from -6.5 to -7.0kcal/mol. To elucidate the binding interactions of the probe with a membrane on the molecular level, a distribution and favorable location of BBP in a POPC bilayer were modeled via atomistic molecular dynamics (MD) simulations using two different approaches: (i) free, diffusion-driven partitioning of the probe molecules into a bilayer and (ii) constrained umbrella sampling of a penetration profile of the dye molecule across a bilayer. Both of these MD approaches agreed with regard to the preferred location of a BBP fluorophore within the interfacial region of a bilayer, located between the hydrocarbon acyl tails and the initial portion of the lipid headgroups. MD simulations also revealed restricted permeability of water molecules into this region of a POPC bilayer, determining the strong fluorescence enhancement observed experimentally for the membrane-partitioned form of BBP.
Copyright © 2010 Elsevier B.V. All rights reserved.
Figures
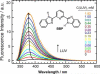


 ), and 25POPC/75POPG (blue △)). The changes in quenching suggest that Co2+ ions do not readily penetrate the hydrophobic region of the lipid vesicle; so that the deeply buried BBP molecules become significantly inaccessible to the quencher. Fluorescence intensities were excited at 315 nm and measured at either 397 nm (free dyes) or 379 nm (in LUV). The quenching experiments in the presence of LUV were carried out at lipid saturation of 2 mM (see Figure 2 for more details).
), and 25POPC/75POPG (blue △)). The changes in quenching suggest that Co2+ ions do not readily penetrate the hydrophobic region of the lipid vesicle; so that the deeply buried BBP molecules become significantly inaccessible to the quencher. Fluorescence intensities were excited at 315 nm and measured at either 397 nm (free dyes) or 379 nm (in LUV). The quenching experiments in the presence of LUV were carried out at lipid saturation of 2 mM (see Figure 2 for more details).


References
-
- Jurkiewicz P, Sýkora J, Olżyńska A, Humpolíčková J, Hof M. Solvent relaxation in phospholipid bilayers: Principles and recent applications. J. Fluorescence. 2005;15:883–894. - PubMed
-
- Bagatolli LA. To see or not to see: Lateral organization of biological membranes and fluorescence microscopy. Biochim. Biophys. Acta. 2006;1758:1541–1556. - PubMed
-
- Kachel K, Asuncion-Punzalan E, London E. The location of fluorescence probes with charged groups in model membranes. Biochim. Biophys. Acta. 1998;1374:63–76. - PubMed
-
- De Vequi-Suplicy CC, Benatti CR, Lamy MT. Laurdan in fluid bilayers: Position and structural sensitivity. J. Fluorescence. 2006;16:431–439. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

