Microtubule disruption targets HIF-1alpha mRNA to cytoplasmic P-bodies for translational repression
- PMID: 21220510
- PMCID: PMC3019555
- DOI: 10.1083/jcb.201004145
Microtubule disruption targets HIF-1alpha mRNA to cytoplasmic P-bodies for translational repression
Abstract
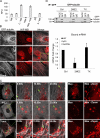
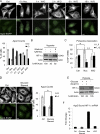
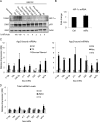
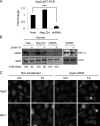
The hypoxia inducible factor 1α (HIF-1α) is overexpressed in solid tumors, driving tumor angiogenesis and survival. However, the mechanisms regulating HIF-1α expression in solid tumors are not fully understood. In this study, we find that microtubule integrity and dynamics are intricately involved in orchestrating HIF-1α translation. HIF-1α messenger RNA (mRNA) traffics on dynamic microtubules when it is actively translated. Microtubule perturbation by taxol (TX) and other microtubule-targeting drugs stalls HIF-1α mRNA transport and releases it from polysomes, suppressing its translation. Immunoprecipitation of the P-body component Argonaute 2 (Ago2) after microtubule disruption shows significant enrichment of HIF-1α mRNAs and HIF-targeting microRNAs (miRNAs). Inhibition of HIF-repressing miRNAs or Ago2 knockdown abrogates TX's ability to suppress HIF-1α translation. Interestingly, microtubule repolymerization after nocodazole washout allows HIF-1α mRNA to reenter active translation, suggesting that microtubule dynamics exert tight yet reversible control over HIF-1α translation. Collectively, we provide evidence for a new mechanism of microtubule-dependent HIF-1α translation with important implications for cell biology.
Figures





References
-
- Anderson P., Kedersha N. 2002. Stressful initiations. J. Cell Sci. 115:3227–3234 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

