Cardiovascular safety of non-steroidal anti-inflammatory drugs: network meta-analysis
- PMID: 21224324
- PMCID: PMC3019238
- DOI: 10.1136/bmj.c7086
Cardiovascular safety of non-steroidal anti-inflammatory drugs: network meta-analysis
Abstract
Objective: To analyse the available evidence on cardiovascular safety of non-steroidal anti-inflammatory drugs.
Design: Network meta-analysis.
Data sources: Bibliographic databases, conference proceedings, study registers, the Food and Drug Administration website, reference lists of relevant articles, and reports citing relevant articles through the Science Citation Index (last update July 2009). Manufacturers of celecoxib and lumiracoxib provided additional data.
Study selection: All large scale randomised controlled trials comparing any non-steroidal anti-inflammatory drug with other non-steroidal anti-inflammatory drugs or placebo. Two investigators independently assessed eligibility.
Data extraction: The primary outcome was myocardial infarction. Secondary outcomes included stroke, death from cardiovascular disease, and death from any cause. Two investigators independently extracted data.
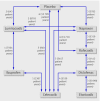
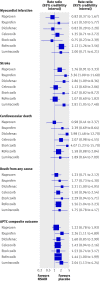
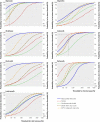
Data synthesis: 31 trials in 116 429 patients with more than 115 000 patient years of follow-up were included. Patients were allocated to naproxen, ibuprofen, diclofenac, celecoxib, etoricoxib, rofecoxib, lumiracoxib, or placebo. Compared with placebo, rofecoxib was associated with the highest risk of myocardial infarction (rate ratio 2.12, 95% credibility interval 1.26 to 3.56), followed by lumiracoxib (2.00, 0.71 to 6.21). Ibuprofen was associated with the highest risk of stroke (3.36, 1.00 to 11.6), followed by diclofenac (2.86, 1.09 to 8.36). Etoricoxib (4.07, 1.23 to 15.7) and diclofenac (3.98, 1.48 to 12.7) were associated with the highest risk of cardiovascular death.
Conclusions: Although uncertainty remains, little evidence exists to suggest that any of the investigated drugs are safe in cardiovascular terms. Naproxen seemed least harmful. Cardiovascular risk needs to be taken into account when prescribing any non-steroidal anti-inflammatory drug.
Conflict of interest statement
Competing interests: All authors have completed the Unified Competing Interest form at
Figures

Comment in
-
Cardiovascular safety of NSAIDs.BMJ. 2011 Jan 11;342:c6618. doi: 10.1136/bmj.c6618. BMJ. 2011. PMID: 21224323 No abstract available.
-
Network meta-analysis. Results can be summarised in a simple figure.BMJ. 2011 Mar 23;342:d1555. doi: 10.1136/bmj.d1555. BMJ. 2011. PMID: 21430004 No abstract available.
-
NSAID use associated with increased cardiovascular risk and death, but naproxen appears to be the least harmful.Evid Based Med. 2011 Oct;16(5):142-3. doi: 10.1136/ebm11033. Epub 2011 Apr 3. Evid Based Med. 2011. PMID: 21460398 No abstract available.
-
Pain: The cardiovascular risks of NSAID use: which NSAID is the safest of them all?Nat Rev Rheumatol. 2011 Mar;7(3):129. doi: 10.1038/nrrheum.2011.9. Nat Rev Rheumatol. 2011. PMID: 21491645 No abstract available.
References
-
- Dai C, Stafford RS, Alexander GC. National trends in cyclooxygenase-2 inhibitor use since market release: nonselective diffusion of a selectively cost-effective innovation. Arch Intern Med 2005;165:171-7. - PubMed
-
- Kaufman DW, Kelly JP, Rosenberg L, Anderson TE, Mitchell AA. Recent patterns of medication use in the ambulatory adult population of the United States: the Slone survey. JAMA 2002;287:337-44. - PubMed
-
- Bresalier RS, Sandler RS, Quan H, Bolognese JA, Oxenius B, Horgan K, et al. Cardiovascular events associated with rofecoxib in a colorectal adenoma chemoprevention trial. N Engl J Med 2005;352:1092-102. - PubMed
-
- Juni P, Nartey L, Reichenbach S, Sterchi R, Dieppe PA, Egger M. Risk of cardiovascular events and rofecoxib: cumulative meta-analysis. Lancet 2004;364:2021-9. - PubMed
-
- McGettigan P, Henry D. Cardiovascular risk and inhibition of cyclooxygenase: a systematic review of the observational studies of selective and nonselective inhibitors of cyclooxygenase 2. JAMA 2006;296:1633-44. - PubMed
