Complete response for advanced liver cancer during sorafenib therapy: case report
- PMID: 21241463
- PMCID: PMC3036648
- DOI: 10.1186/1471-230X-11-4
Complete response for advanced liver cancer during sorafenib therapy: case report
Abstract
Background: Hepatocellular carcinoma (HCC) is the fifth most common neoplasia in the world. In the past, treatment of advanced HCC with conventional antineoplastic drugs did not result in satisfactory outcomes: recently, in this patient population the oral multikinase inhibitor sorafenib has been able to induce a statistically significant improvement of overall survival. Similarly to other anti-angiogenic drugs employed in other tumour types, also sorafenib seldom induces the dimensional tumour shrinking usually observed with conventional cytotoxic drugs: data gathered from studies carried out with sorafenib and other competitors under development do not report any complete response in HCV-induced HCC.
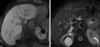
Case presentation: An 84-year old man with a long-lasting history of chronic HCV hepatitis was referred to our Institution for an ultrasonography investigation of a focal hepatic lesion. To better characterize the liver disease and clearly define the diagnosis of the focal hepatic lesion, the patient was hospitalized in our department. Laboratory and instrumental investigations confirmed the clinical picture of HCV-related liver cirrhosis and identified a hepatic lesion of about 6 cm featuring infiltrating HCC with thrombosis of the portal trunk. Due to the advanced stage of the disease, therapy with sorafenib 400 mg bid was started. Right from one month after the treatment was started, a reduction of alpha-fetoprotein level was observed which, by the third month, turned down within the normal limits. In addition the CT scan showed 50% reduction of the neoplastic lesion along with canalization of the portal trunk. At the sixth month the normalization of the alpha-fetoprotein level at the lower limit of normality was confirmed and the MRI showed complete disappearance of the neoplasia. In addition a reduction of a metallo-proteinase serum level was observed. At the twelfth month a further MRI confirmed complete response had been maintained. At present the patient is in a follow-up program to evaluate the duration of the complete response.
Conclusions: This case is worth mentioning since, to the best of our knowledge, it represents the first evidence of complete response to sorafenib in an elderly patient with advanced HCV-related HCC.
Figures




References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

