Angiocidin inhibits breast cancer proliferation through activation of epidermal growth factor receptor and nuclear factor kappa (NF-ĸB)
- PMID: 21241690
- PMCID: PMC3092007
- DOI: 10.1016/j.yexmp.2011.01.002
Angiocidin inhibits breast cancer proliferation through activation of epidermal growth factor receptor and nuclear factor kappa (NF-ĸB)
Abstract
Angiocidin, a tumor-associated peptide, has been previously shown to inhibit tumor progression by blocking angiogenesis. We now show that angiocidin has a direct inhibitory effect on tumor cell proliferation. MDA-MB-231 breast cancer cells were inhibited from proliferating in the presence of epidermal growth factor (EGF) and angiocidin. Angiocidin transfected breast cancer cells also displayed growth inhibition in vitro and failed to develop significant tumors in mice as compared to vector controls. The anti-proliferative effect of angiocidin was reversed by treating the cells with the epidermal growth factor receptor (EGFR) inhibitor 4557W, a potent tyrosine kinase inhibitor. Consistent with these results, we found that treatment of breast cancer cells with angiocidin induced a 2.3 fold increase in EGFR tyrosine 845 phosphorylation while no change in phosphorylation was observed in the remaining 16 phosphorylation sites of EGFR and those of its family members as measured by a human EGFR phosphorylation array. Treatment of breast cancer cells with angiocidin also resulted in the activation of nuclear factor ĸB (Nf-ĸB) and the de novo up-regulation of many down-stream genes transcribed by Nf-ĸB, including cytokines, inflammatory mediators and the cell cycle inhibitor p21(waf1). Therefore, angiocidin is a peptide that not only inhibits tumor angiogenesis but also directly induces inhibition of tumor growth progression through the activation of EGFR and down-stream genes transcribed by Nf-ĸB.
Copyright © 2011 Elsevier Inc. All rights reserved.
Figures




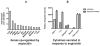
References
-
- Aderem A, Ulevitch RJ. Toll-like receptors in the induction of the innate immune response. Nature. 2000;406:782–7. - PubMed
-
- Biswas DK, Iglehart JD. Linkage between EGFR family receptors and nuclear factor kappaB (NF-kappaB) signaling in breast cancer. J Cell Physiol. 2006;209:645–52. - PubMed
-
- Davis RJ. Signal transduction by the JNK group of MAP kinases. Cell. 2000;103:239–52. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

