Induction of transplantation tolerance converts potential effector T cells into graft-protective regulatory T cells
- PMID: 21243638
- PMCID: PMC3175037
- DOI: 10.1002/eji.201040509
Induction of transplantation tolerance converts potential effector T cells into graft-protective regulatory T cells
Abstract
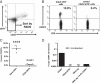
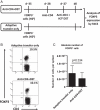
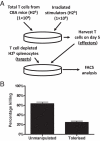
Naturally occurring FOXP3(+) CD4(+) Treg have a crucial role in self-tolerance. The ability to generate similar populations against alloantigens offers the possibility of preventing transplant rejection without indefinite global immunosuppression. Exposure of mice to donor alloantigens combined with anti-CD4 antibody induces operational tolerance to cardiac allografts, and generates Treg that prevent skin and islet allograft rejection in adoptive transfer models. If protocols that generate Treg in vivo are to be developed in the clinical setting it will be important to know the origin of the Treg population and the mechanisms responsible for their generation. In this study, we demonstrate that graft-protective Treg arise in vivo both from naturally occurring FOXP3(+) CD4(+) Treg and from non-regulatory FOXP3(-) CD4(+) cells. Importantly, tolerance induction also inhibits CD4(+) effector cell priming and T cells from tolerant mice have impaired effector function in vitro. Thus, adaptive tolerance induction shapes the immune response to alloantigen by converting potential effector cells into graft-protective Treg and by expanding alloreactive naturally occurring Treg. In relation to clinical tolerance induction, the data indicate that while the generation of alloreactive Treg may be critical for long-term allograft survival without chronic immunosuppression, successful protocols will also require strategies that target potential effector cells.
Copyright © 2011 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
Figures






References
-
- Hariharan S, Johnson CP, Bresnahan BA, Taranto SE, McIntosh MJ, Stablein D. Improved graft survival after renal transplantation in the United States, 1988 to 1996. N. Engl. J. Med. 2000;342:605–612. - PubMed
-
- Meier-Kriesche HU, Schold JD, Srinivas TR, Kaplan B. Lack of improvement in renal allograft survival despite a marked decrease in acute rejection rates over the most recent era. Am. J. Transplant. 2004;4:378–383. - PubMed
-
- Buell JF, Gross TG, Woodle ES. Malignancy after transplantation. Transplantation. 2005;80:S254–S264. - PubMed
-
- Fishman JA. Infection in solid-organ transplant recipients. N. Engl. J. Med. 2007;357:2601–2614. - PubMed
-
- Chatenoud L, Bluestone JA. CD3-specific antibodies: a portal to the treatment of autoimmunity. Nat. Rev. Immunol. 2007;7:622–632. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

