The pharmacokinetic/pharmacodynamic pipeline: translating anticancer drug pharmacology to the clinic
- PMID: 21246315
- PMCID: PMC3032092
- DOI: 10.1208/s12248-011-9253-1
The pharmacokinetic/pharmacodynamic pipeline: translating anticancer drug pharmacology to the clinic
Abstract
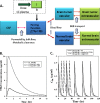
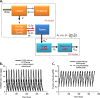
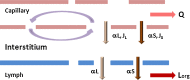
Progress in an understanding of the genetic basis of cancer coupled to molecular pharmacology of potential new anticancer drugs calls for new approaches that are able to address key issues in the drug development process, including pharmacokinetic (PK) and pharmacodynamic (PD) relationships. The incorporation of predictive preclinical PK/PD models into rationally designed early-stage clinical trials offers a promising way to relieve a significant bottleneck in the drug discovery pipeline. The aim of the current review is to discuss some considerations for how quantitative PK and PD analyses for anticancer drugs may be conducted and integrated into a global translational effort, and the importance of examining drug disposition and dynamics in target tissues to support the development of preclinical PK/PD models that can be subsequently extrapolated to predict pharmacologic characteristics in patients. In this article, we describe three different physiologically based (PB) PK modeling approaches, i.e., the whole-body PBPK model, the hybrid PBPK model, and the two-pore model for macromolecules, as well as their applications. General conclusions are that greater effort should be made to generate more clinical data that could validate scaled preclinical PB-PK/PD tumor-based models and, thus, stimulate a framework for preclinical to clinical translation. Finally, given the innovative techniques to measure tissue drug concentrations and associated biomarkers of drug responses, development of predictive PK/PD models will become a standard approach for drug discovery and development.
Figures
Similar articles
-
Translational pharmacokinetic/pharmacodynamic analysis in cancer pharmacology: a tool to maximize the value of antitumor efficacy from tumor bearing mice.Drug Discov Today Technol. 2016 Sep-Dec;21-22:51-56. doi: 10.1016/j.ddtec.2016.08.003. Epub 2016 Oct 21. Drug Discov Today Technol. 2016. PMID: 27978988 Review.
-
Transitioning from Basic toward Systems Pharmacodynamic Models: Lessons from Corticosteroids.Pharmacol Rev. 2020 Apr;72(2):414-438. doi: 10.1124/pr.119.018101. Pharmacol Rev. 2020. PMID: 32123034 Free PMC article. Review.
-
Physiologically based pharmacokinetic and pharmacodynamic modeling in cancer drug development: status, potential and gaps.Expert Opin Drug Metab Toxicol. 2015 May;11(5):743-56. doi: 10.1517/17425255.2015.1037276. Expert Opin Drug Metab Toxicol. 2015. PMID: 25940026 Review.
-
Pharmacokinetic/Pharmacodynamic Modeling for Drug Development in Oncology.Am Soc Clin Oncol Educ Book. 2017;37:210-215. doi: 10.1200/EDBK_180460. Am Soc Clin Oncol Educ Book. 2017. PMID: 28561730 Review.
-
Preclinical pharmacokinetic/pharmacodynamic modeling and simulation in the pharmaceutical industry: an IQ consortium survey examining the current landscape.AAPS J. 2015 Mar;17(2):462-73. doi: 10.1208/s12248-014-9716-2. Epub 2015 Jan 29. AAPS J. 2015. PMID: 25630504 Free PMC article.
Cited by
-
Integrated PK-PD and agent-based modeling in oncology.J Pharmacokinet Pharmacodyn. 2015 Apr;42(2):179-89. doi: 10.1007/s10928-015-9403-7. Epub 2015 Jan 15. J Pharmacokinet Pharmacodyn. 2015. PMID: 25588379 Free PMC article. Review.
-
Non-invasive, real-time reporting drug release in vitro and in vivo.Chem Commun (Camb). 2015 Apr 25;51(32):6948-51. doi: 10.1039/c4cc09920f. Epub 2015 Mar 23. Chem Commun (Camb). 2015. PMID: 25798457 Free PMC article.
-
Phase 1 study of ixazomib, an investigational proteasome inhibitor, in advanced non-hematologic malignancies.Invest New Drugs. 2015 Jun;33(3):652-63. doi: 10.1007/s10637-015-0230-x. Epub 2015 Mar 18. Invest New Drugs. 2015. PMID: 25777468 Free PMC article. Clinical Trial.
-
Translational PK-PD modeling in pain.J Pharmacokinet Pharmacodyn. 2013 Jun;40(3):401-18. doi: 10.1007/s10928-012-9282-0. Epub 2012 Nov 30. J Pharmacokinet Pharmacodyn. 2013. PMID: 23197248 Review.
-
Computational oncology--mathematical modelling of drug regimens for precision medicine.Nat Rev Clin Oncol. 2016 Apr;13(4):242-54. doi: 10.1038/nrclinonc.2015.204. Epub 2015 Nov 24. Nat Rev Clin Oncol. 2016. PMID: 26598946 Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

