Structure and activity of alpha-conotoxin PeIA at nicotinic acetylcholine receptor subtypes and GABA(B) receptor-coupled N-type calcium channels
- PMID: 21252227
- PMCID: PMC3060477
- DOI: 10.1074/jbc.M110.196170
Structure and activity of alpha-conotoxin PeIA at nicotinic acetylcholine receptor subtypes and GABA(B) receptor-coupled N-type calcium channels
Abstract
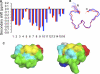
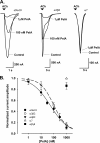
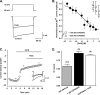
α-Conotoxins are peptides from cone snails that target the nicotinic acetylcholine receptor (nAChR). RgIA and Vc1.1 have analgesic activity in animal pain models. Both peptides target the α9α10 nAChR and inhibit N-type calcium channels via GABA(B) receptor activation, but the mechanism of action of analgesic activity is unknown. PeIA has previously been shown to inhibit the α9α10 and α3β2 nAChRs. In this study, we have determined the structure of PeIA and shown that it is also a potent inhibitor of N-type calcium channels via GABA(B) receptor activation. The characteristic α-conotoxin fold is present in PeIA, but it has a different distribution of surface-exposed hydrophobic and charged residues compared with Vc1.1. Thus, the surface residue distribution, rather than the overall fold, appears to be responsible for the 50-fold increase in selectivity at the α3β2 nAChR by PeIA relative to Vc1.1. In contrast to their difference in potency at the nAChR, the equipotent activity of PeIA and Vc1.1 at the GABA(B) receptor suggests that the GABA(B) receptor is more tolerant to changes in surface residues than is the nAChR. The conserved Asp-Pro-Arg motif of Vc1.1 and RgIA, which is crucial for potency at the α9α10 nAChR, is not required for activity at GABA(B) receptor/N-type calcium channels because PeIA has a His-Pro-Ala motif in the equivalent position. This study shows that different structure-activity relationships are associated with the targeting of the GABA(B) receptor versus nAChRs. Furthermore, there is probably a much more diverse range of conotoxins that target the GABA(B) receptor than currently realized.
Figures

Similar articles
-
Analgesic α-conotoxins Vc1.1 and RgIA inhibit N-type calcium channels in sensory neurons of α9 nicotinic receptor knockout mice.Channels (Austin). 2010 Jan-Feb;4(1):51-4. doi: 10.4161/chan.4.1.10281. Epub 2010 Jan 8. Channels (Austin). 2010. PMID: 20368690
-
Structure-Activity Studies Reveal the Molecular Basis for GABAB-Receptor Mediated Inhibition of High Voltage-Activated Calcium Channels by α-Conotoxin Vc1.1.ACS Chem Biol. 2018 Jun 15;13(6):1577-1587. doi: 10.1021/acschembio.8b00190. Epub 2018 May 25. ACS Chem Biol. 2018. PMID: 29746088
-
Scanning mutagenesis of alpha-conotoxin Vc1.1 reveals residues crucial for activity at the alpha9alpha10 nicotinic acetylcholine receptor.J Biol Chem. 2009 Jul 24;284(30):20275-84. doi: 10.1074/jbc.M109.015339. Epub 2009 May 15. J Biol Chem. 2009. PMID: 19447885 Free PMC article.
-
Analgesic conotoxins: block and G protein-coupled receptor modulation of N-type (Ca(V) 2.2) calcium channels.Br J Pharmacol. 2012 May;166(2):486-500. doi: 10.1111/j.1476-5381.2011.01781.x. Br J Pharmacol. 2012. PMID: 22091786 Free PMC article. Review.
-
Conotoxin Interactions with α9α10-nAChRs: Is the α9α10-Nicotinic Acetylcholine Receptor an Important Therapeutic Target for Pain Management?Toxins (Basel). 2015 Sep 28;7(10):3916-32. doi: 10.3390/toxins7103916. Toxins (Basel). 2015. PMID: 26426047 Free PMC article. Review.
Cited by
-
α7- and α9-Containing Nicotinic Acetylcholine Receptors in the Functioning of Immune System and in Pain.Int J Mol Sci. 2023 Mar 30;24(7):6524. doi: 10.3390/ijms24076524. Int J Mol Sci. 2023. PMID: 37047495 Free PMC article. Review.
-
αD-Conotoxins in Species of the Eastern Pacific: The Case of Conus princeps from Mexico.Toxins (Basel). 2019 Jul 12;11(7):405. doi: 10.3390/toxins11070405. Toxins (Basel). 2019. PMID: 31336928 Free PMC article.
-
Synthesis of Cystine-Stabilised Dicarba Conotoxin EpI: Ring-Closing Metathesis of Sidechain Deprotected, Sulfide-Rich Sequences.Mar Drugs. 2023 Jun 29;21(7):390. doi: 10.3390/md21070390. Mar Drugs. 2023. PMID: 37504921 Free PMC article.
-
α-Conotoxin PeIA[S9H,V10A,E14N] potently and selectively blocks α6β2β3 versus α6β4 nicotinic acetylcholine receptors.Mol Pharmacol. 2012 Nov;82(5):972-82. doi: 10.1124/mol.112.080853. Epub 2012 Aug 22. Mol Pharmacol. 2012. PMID: 22914547 Free PMC article.
-
Cloning, Synthesis and Functional Characterization of a Novel α-Conotoxin Lt1.3.Mar Drugs. 2018 Mar 31;16(4):112. doi: 10.3390/md16040112. Mar Drugs. 2018. PMID: 29614714 Free PMC article.
References
-
- McIntosh J. M., Gardner S., Luo S., Garrett J. E., Yoshikami D. (2000) Eur. J. Pharmacol. 393, 205–208 - PubMed
-
- Dutton J. L., Craik D. J. (2001) Curr. Med. Chem. 8, 327–344 - PubMed
-
- Terlau H., Olivera B. M. (2004) Physiol. Rev. 84, 41–68 - PubMed
-
- Millard E. L., Daly N. L., Craik D. J. (2004) Eur. J. Biochem. 271, 2320–2326 - PubMed
-
- Hu S. H., Gehrmann J., Alewood P. F., Craik D. J., Martin J. L. (1997) Biochemistry 36, 11323–11330 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

