Vaccine development against Neisseria meningitidis
- PMID: 21255369
- PMCID: PMC3815792
- DOI: 10.1111/j.1751-7915.2010.00178.x
Vaccine development against Neisseria meningitidis
Abstract
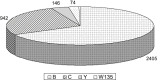
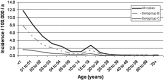
Meningococcal disease is communicable by close contact or droplet aerosols. Striking features are high case fatality rates and peak incidences of invasive disease in infants, toddlers and adolescents. Vaccine development is hampered by bacterial immune evasion strategies including molecular mimicry.As for Haemophilus influenzae and Streptococcus pneumoniae, no vaccine has therefore been developed that targets all serogroups of Neisseria meningitidis. Polysaccharide vaccines available both in protein conjugated and non-conjugated form, have been introduced against capsular serogroups A, C,W-135 and Y, but are ineffective against serogroup B meningococci, which cause a significant burden of disease in many parts of the world. Detoxified outer membrane vesicles are used since decades to elicit protection against epidemic serogroup B disease. Genome mining and biochemical approaches have provided astounding progress recently in the identification of immunogenic, yet reasonably conserved outer membrane proteins. As subcapsular proteins nevertheless are unlikely to immunize against all serogroup B variants, thorough investigation by surrogate assays and molecular epidemiology approaches are needed prior to introduction and post-licensure of protein vaccines. Research currently addresses the analysis of life vaccines, meningococcus B polysaccharide modifications and mimotopes, as well as the use of N. lactamica outer membrane vesicles.
Figures

References
-
- Achtman M., Wagner M. Microbial diversity and the genetic nature of microbial species. Nat Rev Microbiol. 2008;6:431–440. - PubMed
-
- van Alphen L., van den Dobbelsteen G. Meningococcal B vaccine development and evaluation of efficacy. Hum Vaccin. 2008;4:158–161. - PubMed
-
- Ashton F.E., Ryan J.A., Michon F., Jennings H.J. Protective efficacy of mouse serum to the N‐propionyl derivative of meningococcal group B polysaccharide. Microb Pathog. 1989;6:455–458. - PubMed
-
- Auckland C., Gray S., Borrow R., Andrews N., Goldblatt D., Ramsay M., Miller E. Clinical and immunologic risk factors for meningococcal C conjugate vaccine failure in the United Kingdom. J Infect Dis. 2006;194:1745–1752. - PubMed
-
- Balmer P., Borrow R., Miller E. Impact of meningococcal C conjugate vaccine in the UK. J Med Microbiol. 2002;51:717–722. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

