Proinflammatory role of aquaporin-4 in autoimmune neuroinflammation
- PMID: 21257712
- PMCID: PMC3079299
- DOI: 10.1096/fj.10-177279
Proinflammatory role of aquaporin-4 in autoimmune neuroinflammation
Abstract
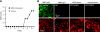
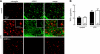
Aquaporin-4 (AQP4) deficiency in mice reduces neuroinflammation in experimental autoimmune encephalomyelitis (EAE) produced by active immunization with myelin oligodendrocyte glycoprotein peptide (MOG). Potential mechanisms for the protective effect of AQP4 deficiency were investigated, including AQP4-dependent leukocyte and microglia cell function, immune cell entry in the central nervous system (CNS), intrinsic neuroinflammation, and humoral immune response. As we found with active-immunization EAE, neuroinflammation was greatly reduced in AQP4-knockout mice in adoptive-transfer EAE. AQP4 was absent in immune cells, including activated T lymphocytes. The CNS migration of fluorescently labeled, MOG-sensitized T lymphocytes was comparable in wild-type and AQP4-knockout mice. Microglia did not express AQP4. Serum anti-AQP4 antibodies were absent in EAE. Remarkably, intracerebral injection of LPS produced much greater neuroinflammation in wild-type than in AQP4-knockout mice, and cytokine (TNF-α and IL-6) secretion was reduced in astrocyte cultures from AQP4-knockout mice. Adenovirus-mediated expression of AQP4, or of an unrelated aquaporin, AQP1, increased cytokine secretion in astrocyte and nonastrocyte cell cultures, supporting the involvement of aquaporin water permeability in cytokine secretion. Our data suggest an intrinsic proinflammatory role of AQP4 involving AQP4-dependent astrocyte swelling and cytokine release. Reduction in AQP4 water transport may be protective in neuroinflammatory CNS diseases.
Figures


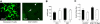


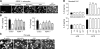
References
-
- Korn T., Bettelli E., Oukka M., Kuchroo V. K. (2009) IL-17 and Th17 cells. Annu. Rev. Immunol. 27, 485–517 - PubMed
-
- Lyons J. A., Ramsbottom M. J., Cross A. H. (2002) Critical role of antigen-specific antibody in experimental autoimmune encephalomyelitis induced by recombinant myelin oligodendrocyte glycoprotein. Eur. J. Immunol. 32, 1905–1913 - PubMed
-
- Weber M. S., Prod'homme T., Patarroyo J. C., Molnarfi N., Karnezis T., Lehmann-Horn K., Danilenko D. M., Eastham-Anderson J., Slavin A. J., Linington C., Bernard C. C., Martin F., Zamvil S. S. (2010) B-cell activation influences T-cell polarization and outcome of anti-CD20 B-cell depletion in central nervous system autoimmunity. Ann. Neurol. 68, 369–383 - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 EY013574/EY/NEI NIH HHS/United States
- DK86125/DK/NIDDK NIH HHS/United States
- R01 EB000415/EB/NIBIB NIH HHS/United States
- R01 DK035124/DK/NIDDK NIH HHS/United States
- AI73737/AI/NIAID NIH HHS/United States
- EY13574/EY/NEI NIH HHS/United States
- DK35124/DK/NIDDK NIH HHS/United States
- R01 NS063008/NS/NINDS NIH HHS/United States
- HL73856/HL/NHLBI NIH HHS/United States
- NS63008/NS/NINDS NIH HHS/United States
- R01 HL073856/HL/NHLBI NIH HHS/United States
- P30 DK072517/DK/NIDDK NIH HHS/United States
- DK72517/DK/NIDDK NIH HHS/United States
- R01 AI073737/AI/NIAID NIH HHS/United States
- RC1 DK086125/DK/NIDDK NIH HHS/United States
- EB00415/EB/NIBIB NIH HHS/United States
- R37 DK035124/DK/NIDDK NIH HHS/United States
- R37 EB000415/EB/NIBIB NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

