Suppression of the proinflammatory response of metastatic melanoma cells increases TRAIL-induced apoptosis
- PMID: 21268068
- PMCID: PMC4378843
- DOI: 10.1002/jcb.22934
Suppression of the proinflammatory response of metastatic melanoma cells increases TRAIL-induced apoptosis
Abstract
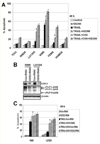
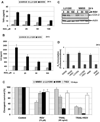
Melanoma is the most lethal form of human skin cancer. However, only limited chemotherapy is currently available for the metastatic stage of the disease. Since chemotherapy, radiation and sodium arsenite treatment operate mainly through induction of the intrinsic mitochondrial pathway, a strongly decreased mitochondrial function in metastatic melanoma cells, could be responsible for low efficacy of the conventional therapy of melanoma. Another feature of metastatic melanoma cells is their proinflammatory phenotype, linked to endogenous expression of the inflammatory cytokines, such as TNFα IL6 and IL8, their receptors, and constitutive NF-κB- and STAT3-dependent gene expression, including cyclooxygenase-2 (PTGS2/COX2). In the present study, we treated melanoma cells with immunological (monoclonal antibody against TNFα or IL6), pharmacological (small molecular inhibitors of IKKβ-NF-κB and JAK2-STAT3) or genetic (specific RNAi for COX-2) agents that suppressed the inflammatory response in combination with induction of apoptosis via TRAIL. As a result of these combined treatments, exogenous TRAIL via interactions with TRAIL-R2/R1 strongly increased levels of apoptosis in resistant melanoma cells. The present study provides new understanding of the regulation of TRAIL-mediated apoptosis in melanoma and will serve as the foundation for the potential development of a novel approach for a therapy of resistant melanomas.
Copyright © 2010 Wiley-Liss, Inc.
Figures





References
-
- Aggarwal BB, Bhardwaj A, Aggarwal RS, Seeram NP, Shishodia S, Takada Y. Role of resveratrol in prevention and therapy of cancer: Preclinical and clinical studies. Anticancer Res. 2004;24:2783–2840. - PubMed
-
- Alexeyev MF, Ledoux SP, Wilson GL. Mitochondrial DNA and aging. Clin Sci (Lond) 2004;107:355–364. - PubMed
-
- Amadori S, Fenaux P, Ludwig H, O’Dwyer M, Sanz M. Use of arsenic trioxide in haematological malignancies: Insight into the clinical development of a novel agent. Curr Med Res Opin. 2005;21:403–411. - PubMed
-
- Ashkenazi A, Holland P, Eckhardt SG. Ligand-based targeting of apoptosis in cancer: The potential of recombinant human apoptosis ligand 2/Tumor necrosis factor-related apoptosis-inducing ligand (rhApo2L/ TRAIL) J Clin Oncol. 2008;26:3621–3630. - PubMed
-
- Beal MF. Mitochondrial dysfunction and oxidative damage in Alzheimer’s and Parkinson’s diseases and coenzyme Q10 as a potential treatment. J Bioenerg Biomembr. 2004;36:381–386. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

