Insulin degludec in type 1 diabetes: a randomized controlled trial of a new-generation ultra-long-acting insulin compared with insulin glargine
- PMID: 21270174
- PMCID: PMC3041203
- DOI: 10.2337/dc10-1925
Insulin degludec in type 1 diabetes: a randomized controlled trial of a new-generation ultra-long-acting insulin compared with insulin glargine
Abstract
Objective: Insulin degludec (IDeg) is a basal insulin that forms soluble multihexamers after subcutaneous injection, resulting in an ultra-long action profile. We assessed the efficacy and safety of IDeg formulations administered once daily in combination with mealtime insulin aspart in people with type 1 diabetes.
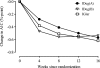
Research design and methods: In this 16-week, randomized, open-label trial, participants (mean: 45.8 years old, A1C 8.4%, fasting plasma glucose [FPG] 9.9 mmol/L, BMI 26.9 kg/m(2)) received subcutaneous injections of IDeg(A) (600 μmol/L; n = 59), IDeg(B) (900 μmol/L; n = 60), or insulin glargine (IGlar; n = 59), all given once daily in the evening. Insulin aspart was administered at mealtimes. RESULTS At 16 weeks, mean A1C was comparable for IDeg(A) (7.8 ± 0.8%), IDeg(B) (8.0 ± 1.0%), and IGlar (7.6 ± 0.8%), as was FPG (8.3 ± 4.0, 8.3 ± 2.8, and 8.9 ± 3.5 mmol/L, respectively). Estimated mean rates of confirmed hypoglycemia were 28% lower for IDeg(A) compared with IGlar (rate ratio [RR]: 0.72 [95% CI 0.52-1.00]) and 10% lower for IDeg(B) compared with IGlar (RR: 0.90 [0.65-1.24]); rates of nocturnal hypoglycemia were 58% lower for IDeg(A) (RR: 0.42 [0.25-0.69]) and 29% lower for IDeg(B) (RR: 0.71 [0.44-1.16]). Mean total daily insulin dose was similar to baseline. The frequency and pattern of adverse events was similar between insulin treatments.
Conclusions: In this clinical exploratory phase 2 trial in people with type 1 diabetes, IDeg is safe and well tolerated and provides comparable glycemic control to IGlar at similar doses, with reduced rates of hypoglycemia.
Trial registration: ClinicalTrials.gov NCT00612040.
Figures

References
-
- The Diabetes Control and Complications Trial Research Group The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med 1993;329:977–986 - PubMed
-
- Nathan DM, Cleary PA, Backlund JY, et al. Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications (DCCT/EDIC) Study Research Group Intensive diabetes treatment and cardiovascular disease in patients with type 1 diabetes. N Engl J Med 2005;353:2643–2653 - PMC - PubMed
-
- The Diabetes Control and Complications Trial Research Group Hypoglycemia in the diabetes control and complications trial. Diabetes 1997;46:271–286 - PubMed
-
- Bartley PC, Bogoev M, Larsen J, Philotheou A. Long-term efficacy and safety of insulin detemir compared to Neutral Protamine Hagedorn insulin in patients with type 1 diabetes using a treat-to-target basal-bolus regimen with insulin aspart at meals: a 2-year, randomized, controlled trial. Diabet Med 2008;25:442–449 - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

