Synaptic release at mammalian bipolar cell terminals
- PMID: 21272392
- PMCID: PMC3157312
- DOI: 10.1017/S0952523810000453
Synaptic release at mammalian bipolar cell terminals
Abstract
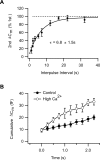
Bipolar cells play a vital role in the transfer of visual information across the vertebrate retina. The synaptic output of these neurons is regulated by factors that are extrinsic and intrinsic. Relatively little is known about the intrinsic factors that regulate neurotransmitter exocytosis. Much of what we know about intrinsic presynaptic mechanisms that regulate glutamate release has come from the study of the unusually large and accessible synaptic terminal of the goldfish rod-dominant bipolar cell, the Mb1 bipolar cell. However, over the past several years, examination of presynaptic mechanisms governing neurotransmitter release has been extended to the mammalian rod bipolar cell. In this review, we discuss the recent advances in our understanding of synaptic vesicle dynamics and neurotransmitter release in rodent rod bipolar cells and consider how these properties help to shape the synaptic output of the mammalian retina.
Figures

Similar articles
-
Syntaxin 3B is essential for the exocytosis of synaptic vesicles in ribbon synapses of the retina.Neuroscience. 2010 Mar 31;166(3):832-41. doi: 10.1016/j.neuroscience.2009.12.075. Epub 2010 Jan 6. Neuroscience. 2010. PMID: 20060037 Free PMC article.
-
Global Ca2+ signaling drives ribbon-independent synaptic transmission at rod bipolar cell synapses.J Neurosci. 2014 Apr 30;34(18):6233-44. doi: 10.1523/JNEUROSCI.5324-13.2014. J Neurosci. 2014. PMID: 24790194 Free PMC article.
-
Synaptic mechanisms of bipolar cell terminals.Vision Res. 1999 Jul;39(15):2469-76. doi: 10.1016/s0042-6989(98)00249-1. Vision Res. 1999. PMID: 10396616
-
Pre- and postsynaptic mechanisms of spontaneous, excitatory postsynaptic currents in the salamander retina.Prog Brain Res. 2001;131:241-53. doi: 10.1016/s0079-6123(01)31020-8. Prog Brain Res. 2001. PMID: 11420944 Review. No abstract available.
-
Optical monitoring of synaptic vesicle trafficking in ribbon synapses.Neurochem Int. 2002 Nov;41(5):307-12. doi: 10.1016/s0197-0186(02)00015-3. Neurochem Int. 2002. PMID: 12176071 Review.
Cited by
-
Phosphatidic acid and neurotransmission.Adv Biol Regul. 2017 Jan;63:15-21. doi: 10.1016/j.jbior.2016.09.004. Epub 2016 Sep 20. Adv Biol Regul. 2017. PMID: 27671966 Free PMC article. Review.
-
Neuromodulator-evoked synaptic metaplasticity within a central pattern generator network.J Neurophysiol. 2012 Nov;108(10):2846-56. doi: 10.1152/jn.00586.2012. Epub 2012 Aug 29. J Neurophysiol. 2012. PMID: 22933725 Free PMC article.
-
Postsynaptic Plasticity Triggered by Ca²⁺-Permeable AMPA Receptor Activation in Retinal Amacrine Cells.Neuron. 2016 Feb 3;89(3):507-20. doi: 10.1016/j.neuron.2015.12.028. Epub 2016 Jan 21. Neuron. 2016. PMID: 26804991 Free PMC article.
-
Phosphorylation of the Retinal Ribbon Synapse Specific t-SNARE Protein Syntaxin3B Is Regulated by Light via a Ca2 +-Dependent Pathway.Front Cell Neurosci. 2020 Oct 20;14:587072. doi: 10.3389/fncel.2020.587072. eCollection 2020. Front Cell Neurosci. 2020. PMID: 33192329 Free PMC article.
-
On optimal coupling of the 'electronic photoreceptors' into the degenerate retina.J Neural Eng. 2020 Jul 24;17(4):045008. doi: 10.1088/1741-2552/aba0d2. J Neural Eng. 2020. PMID: 32613948 Free PMC article.
References
-
- Abercrombie M. Estimation of nuclear population from microtome sections. Anat Rec (Hoboken) 1946;94:239–247. - PubMed
-
- Alexander KR, Fishman GA, Peachey NS, Marchese AL, Tso MO. `On' response defect in paraneoplastic night blindness with cutaneous malignant melanoma. Invest Ophthalmol Vis Sci. 1992;33:477–483. - PubMed
-
- Baumann L, Gerstner A, Zong X, Biel M, Wahl-Schott C. Functional characterization of the L-type Ca2+ channel Cav1.4alpha1 from mouse retina. Invest Ophthalmol Vis Sci. 2004;45:708–713. - PubMed
-
- Bech-Hansen NT, Naylor MJ, Maybaum TA, Pearce WG, Koop B, Fishman GA, Mets M, Musarella MA, Boycott KM. Loss-of-function mutations in a calcium-channel alpha1-subunit gene in Xp11.23 cause incomplete X-linked congenital stationary night blindness. Nat Genet. 1998;19:264–267. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

