Nonlinear effects of noradrenergic modulation of olfactory bulb function in adult rodents
- PMID: 21273323
- PMCID: PMC3075300
- DOI: 10.1152/jn.00960.2010
Nonlinear effects of noradrenergic modulation of olfactory bulb function in adult rodents
Abstract
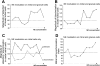
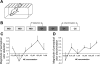
The mammalian main olfactory bulb receives a significant noradrenergic input from the locus coeruleus. Norepinephrine (NE) is involved in acquisition of conditioned odor preferences in neonatal animals, in some species-specific odor-dependent behaviors, and in adult odor perception. We provide a detailed review of the functional role of NE in adult rodent main olfactory bulb function. We include cellular, synaptic, network, and behavioral data and use computational simulations to tie these different types of data together.
Figures




References
-
- Aston-Jones G, Rajkowski J, Cohen J. Locus coeruleus and regulation of behavioral flexibility and attention. Prog Brain Res 126: 165–182, 2000 - PubMed
-
- Aungst JL, Heyward PM, Puche AC, Karnup SV, Hayar A, Szabo G, Shipley MT. Centre-surround inhibition among olfactory bulb glomeruli. Nature 426: 623–629, 2003 - PubMed
-
- Berridge CW, Waterhouse BD. The locus coeruleus-noradrenergic system: modulation of behavioral state and state-dependent cognitive processes. Brain Res Brain Res Rev 42: 33–84, 2003 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

