Concerted action of CB1 cannabinoid receptor and deleted in colorectal cancer in axon guidance
- PMID: 21273433
- PMCID: PMC6623605
- DOI: 10.1523/JNEUROSCI.4134-09.2011
Concerted action of CB1 cannabinoid receptor and deleted in colorectal cancer in axon guidance
Abstract
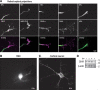
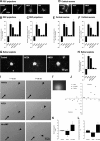
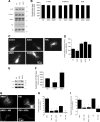
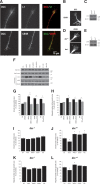
Endocannabinoids (eCBs) are retrograde neurotransmitters that modulate the function of many types of synapses. The presence of eCBs, their CB1 receptor (CB1R), and metabolizing enzymes at embryonic and early postnatal periods have been linked to developmental processes such as neuronal proliferation, differentiation, and migration, axon guidance, and synaptogenesis. Here, we demonstrate the presence of a functional eCB system in the developing visual system and the role of CB1R during axon growth and retinothalamic development. Pharmacological treatment of retinal explants and primary cortical neuron cultures with ACEA, a selective CB1R agonist, induced a collapse of the growth cone (GC). Furthermore the application of AM251, a CB1R inverse agonist, to the neuronal cultures increased the surface area of GC. In vivo, intraocular injection of ACEA diminished retinal projection growth, while AM251 promoted growth and caused aberrant projections. In addition, compared with their wild-type littermates, CB1R-deficient adult mice revealed a lower level of eye-specific segregation of retinal projections in the dorsal lateral geniculate nucleus. Finally, we found that pharmacological modulation of CB1R affected the trafficking of Deleted in colorectal cancer (DCC) receptor to the plasma membrane in a PKA-dependent manner. Moreover, pharmacological inhibition or genetic inactivation of DCC abolished the CB1R-induced reorganization of the GC. Overall, these findings establish a mechanism by which the CB1R influences GC behavior and nervous system development in concerted action with DCC.
Figures
References
-
- Argaw A, Duff G, Boire D, Ptito M, Bouchard J-F. Protein kinase A modulates retinal ganglion cell growth during development. Exp Neurol. 2008;211:494–502. - PubMed
-
- Argaw A, Duff G, Tea N, Ptito M, Bouchard J-F. The endocannabinoid system modulates axon guidance and target selection during development. Soc Neurosci Abstr. 2009;35:129–8.
-
- Barres BA, Silverstein BE, Corey DP, Chun LL. Immunological, morphological, and electrophysiological variation among retinal ganglion cells purified by panning. Neuron. 1988;1:791–803. - PubMed
-
- Berghuis P, Rajnicek AM, Morozov YM, Ross RA, Mulder J, Urbán GM, Monory K, Marsicano G, Matteoli M, Canty A, Irving AJ, Katona I, Yanagawa Y, Rakic P, Lutz B, Mackie K, Harkany T. Hardwiring the brain: endocannabinoids shape neuronal connectivity. Science. 2007;316:1212–1216. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous
