A pronociceptive role for the 5-HT2 receptor on spinal nociceptive transmission: an in vivo electrophysiological study in the rat
- PMID: 21276431
- PMCID: PMC3142932
- DOI: 10.1016/j.brainres.2011.01.057
A pronociceptive role for the 5-HT2 receptor on spinal nociceptive transmission: an in vivo electrophysiological study in the rat
Abstract
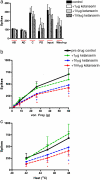
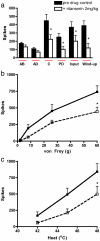
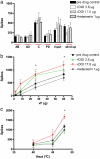
Serotonin (5-HT) plays a major yet complex role in modulating spinal nociceptive transmission as a consequence of the number of 5-HT receptor subtypes. These include the 5-HT2 receptor, which is further sub classified into 5-HT2A, B and C. Studies have described both a pro- and antinociceptive action following 5-HT2A-receptor activation; therefore, to shed light on the directional nature of spinal 5-HT2A receptor activity, we investigated the effects of spinal administration of the 5-HT2A receptor antagonist, ketanserin, on the evoked responses of dorsal horn neurones to electrical, mechanical and thermal stimulation. We also assessed the effects of systemic administration of ritanserin, a 5-HT2A/2C receptor antagonist and spinal application of (±)-2,5-Dimethoxy-4-iodoamphetamine hydrochloride (DOI) (3.6 and 17.8μg/50μl), a 5-HT2A/2C agonist, on the same evoked neuronal responses. Ketanserin (1, 10 and 100μg/50μl) produced a dose related inhibition of the evoked responses to noxious mechanical punctate and thermal stimuli only. Ritanserin (2mg/kg) replicated the inhibitory effects seen with ketanserin on the natural evoked neuronal responses and also potently inhibited the C-fibre, post discharge, input and wind-up evoked responses. DOI increased the mechanical and thermal evoked responses, an effect reversed by ketanserin. Thus, our findings show that spinal ketanserin (1-100μg/50μl) and systemic ritanserin (2mg/kg), at these doses, have similar antinociceptive effects, whereas the agonist, DOI, produced excitatory effects, on spinal neuronal activity. Our data, therefore, supports a pronociceptive role for 5-HT2 receptors, most likely through modulation of 5-HT2A receptor activity, on spinal nociceptive transmission under normal conditions.
Copyright © 2010 Elsevier B.V. All rights reserved.
Figures
Similar articles
-
Activation of spinal serotonin(2A/2C) receptors augments nociceptive responses in the rat.Brain Res. 2001 Aug 10;910(1-2):179-81. doi: 10.1016/s0006-8993(01)02652-x. Brain Res. 2001. PMID: 11489268
-
Effects of 5-HT2 and 5-HT3 receptors on the modulation of nociceptive transmission in rat spinal cord according to the formalin test.Eur J Pharmacol. 2001 Jul 13;424(1):45-52. doi: 10.1016/s0014-2999(01)01117-7. Eur J Pharmacol. 2001. PMID: 11470259
-
Pharmacological characterization of in vivo properties of putative mixed 5-HT1A agonist/5-HT(2A/2C) antagonist anxiolytics. II. Drug discrimination and behavioral observation studies in rats.J Pharmacol Exp Ther. 1997 Aug;282(2):747-59. J Pharmacol Exp Ther. 1997. PMID: 9262338
-
Differential effects of activation of lumbar and thoracic 5-HT2A/2C receptors on nociception in rats.Pharmacol Biochem Behav. 1997 Mar;56(3):523-7. doi: 10.1016/s0091-3057(96)00296-1. Pharmacol Biochem Behav. 1997. PMID: 9077592
-
Theophylline-induced respiratory recovery following cervical spinal cord hemisection is augmented by serotonin 2 receptor stimulation.Brain Res. 2002 Nov 22;956(1):1-13. doi: 10.1016/s0006-8993(02)03097-4. Brain Res. 2002. PMID: 12426040
Cited by
-
Adjunctive effect of the serotonin 5-HT2C receptor agonist lorcaserin on opioid-induced antinociception in mice.Neuropharmacology. 2020 May 1;167:107949. doi: 10.1016/j.neuropharm.2020.107949. Epub 2020 Jan 24. Neuropharmacology. 2020. PMID: 31987863 Free PMC article.
-
Modality selective roles of pro-nociceptive spinal 5-HT2A and 5-HT3 receptors in normal and neuropathic states.Neuropharmacology. 2018 Dec;143:29-37. doi: 10.1016/j.neuropharm.2018.09.028. Epub 2018 Sep 18. Neuropharmacology. 2018. PMID: 30240783 Free PMC article.
-
Pronociceptive and Antinociceptive Effects of Buprenorphine in the Spinal Cord Dorsal Horn Cover a Dose Range of Four Orders of Magnitude.J Neurosci. 2015 Jul 1;35(26):9580-94. doi: 10.1523/JNEUROSCI.0731-14.2015. J Neurosci. 2015. PMID: 26134641 Free PMC article.
-
Serotonergic Modulation of Nociceptive Circuits in Spinal Cord Dorsal Horn.Curr Neuropharmacol. 2019;17(12):1133-1145. doi: 10.2174/1570159X17666191001123900. Curr Neuropharmacol. 2019. PMID: 31573888 Free PMC article. Review.
-
Monoaminergic and Opioidergic Modulation of Brainstem Circuits: New Insights Into the Clinical Challenges of Pain Treatment?Front Pain Res (Lausanne). 2021 Jul 5;2:696515. doi: 10.3389/fpain.2021.696515. eCollection 2021. Front Pain Res (Lausanne). 2021. PMID: 35295506 Free PMC article. Review.
References
-
- Aira Z., Buesa I., Salgueiro M., Bilbao J., Aguilera L., Zimmermann M., Azkue J.J. Subtype-specific changes in 5-HT receptor-mediated modulation of C fibre-evoked spinal field potentials are triggered by peripheral nerve injury. Neuroscience. 2010;168:831–841. - PubMed
-
- Aoki J., Hayashida M., Tagami M., Nagashima M., Fukuda K., Nishizawa D., Ogai Y., Kasai S., Ikeda K., Iwahashi K. Association between 5-hydroxytryptamine 2A receptor gene polymorphism and postoperative analgesic requirements after major abdominal surgery. Neurosci. Lett. 2010;479:40–43. - PubMed
-
- Bondy B., Spaeth M., Offenbaecher M., Glatzeder K., Stratz T., Schwarz M., de Jonge S., Kruger M., Engel R.R., Farber L., Pongratz D.E., Ackenheil M. The T102C polymorphism of the 5-HT2A-receptor gene in fibromyalgia. Neurobiol. Dis. 1999;6:433–439. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

