Molecular annotation of integrative feeding neural circuits
- PMID: 21284989
- PMCID: PMC3286830
- DOI: 10.1016/j.cmet.2010.12.013
Molecular annotation of integrative feeding neural circuits
Abstract
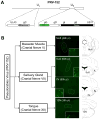
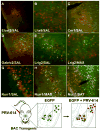
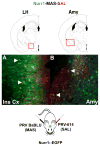
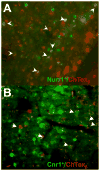
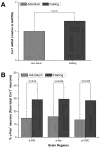
The identity of higher-order neurons and circuits playing an associative role to control feeding is unknown. We injected pseudorabies virus, a retrograde tracer, into masseter muscle, salivary gland, and tongue of BAC-transgenic mice expressing GFP in specific neural populations and identified several CNS regions that project multisynaptically to the periphery. MCH and orexin neurons were identified in the lateral hypothalamus, and Nurr1 and Cnr1 in the amygdala and insular/rhinal cortices. Cholera toxin β tracing showed that insular Nurr1(+) and Cnr1(+) neurons project to the amygdala or lateral hypothalamus, respectively. Finally, we show that cortical Cnr1(+) neurons show increased Cnr1 mRNA and c-Fos expression after fasting, consistent with a possible role for Cnr1(+) neurons in feeding. Overall, these studies define a general approach for identifying specific molecular markers for neurons in complex neural circuits. These markers now provide a means for functional studies of specific neuronal populations in feeding or other complex behaviors.
Copyright © 2011 Elsevier Inc. All rights reserved.
Figures


References
-
- Akamatsu W, Okano HJ, Osumi N, Inoue T, Nakamura S, Sakakibara S, Miura M, Matsuo N, Darnell RB, Okano H. Mammalian ELAV-like neuronal RNA-binding proteins HuB and HuC promote neuronal development in both the central and the peripheral nervous systems. Proc Natl Acad Sci USA. 1999;96:9885–9890. - PMC - PubMed
-
- Anderson DJ, Hector MP. Periodontal mechanoreceptors and parotid secretion in animals and man. J Dent Res. 1987;66:518–523. - PubMed
-
- Cahoy JD, Emery B, Kaushal A, Foo LC, Zamanian JL, Christopherson KS, Xing Y, Lubischer JL, Krieg PA, Krupenko SA, et al. A transcriptome database for astrocytes, neurons, and oligodendrocytes: a new resource for understanding brain development and function. J Neurosci. 2008;28:264–278. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

