Improving adoptive T cell therapy by targeting and controlling IL-12 expression to the tumor environment
- PMID: 21285960
- PMCID: PMC3070103
- DOI: 10.1038/mt.2010.313
Improving adoptive T cell therapy by targeting and controlling IL-12 expression to the tumor environment
Abstract
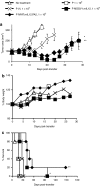
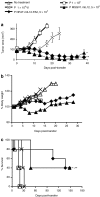
Interleukin-12 (IL-12) is an important immunostimulatory cytokine, yet its clinical application has been limited by the systemic toxicity associated with its administration. In this work, we developed a strategy to selectively deliver IL-12 to the tumor environment using genetically engineered lymphocytes. However, peripheral blood lymphocytes (PBLs) transduced with a γ-retroviral vector, which constitutively expressed IL-12, failed to expand in culture due to apoptosis. To circumvent this problem, a vector was designed where IL-12 expression was directed by a composite promoter-containing binding motifs for nuclear factor of activated T-cells (NFAT.hIL12.PA2). The NFAT-responsive promoter was activated to drive IL-12 expression upon the recognition of tumor-specific antigen mediated by a T cell receptor (TCR) that was engineered into the same lymphocytes. We tested the efficacy of the inducible IL-12 vector in vivo in a murine melanoma model. Adoptive transfer of pmel-1 T cells genetically engineered with NFAT-murineIL12 (NFAT.mIL12.PA2) significantly enhanced regression of large established B16 melanoma. Notably, this targeted and controlled IL-12 treatment was without toxicity. Taken together, our results suggest that using the NFAT.hIL12.PA2 vector might be a promising approach to enhance adoptive cancer immunotherapy.
Figures

References
-
- Gately MK. Interleukin-12: a recently discovered cytokine with potential for enhancing cell-mediated immune responses to tumors. Cancer Invest. 1993;11:500–506. - PubMed
-
- Mehrotra PT, Wu D, Crim JA, Mostowski HS., and, Siegel JP. Effects of IL-12 on the generation of cytotoxic activity in human CD8+ T lymphocytes. J Immunol. 1993;151:2444–2452. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

