A functionally characterized test set of human induced pluripotent stem cells
- PMID: 21293464
- PMCID: PMC3229307
- DOI: 10.1038/nbt.1783
A functionally characterized test set of human induced pluripotent stem cells
Abstract
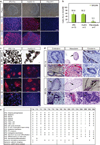
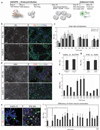
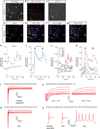
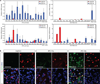
Human induced pluripotent stem cells (iPSCs) present exciting opportunities for studying development and for in vitro disease modeling. However, reported variability in the behavior of iPSCs has called their utility into question. We established a test set of 16 iPSC lines from seven individuals of varying age, sex and health status, and extensively characterized the lines with respect to pluripotency and the ability to terminally differentiate. Under standardized procedures in two independent laboratories, 13 of the iPSC lines gave rise to functional motor neurons with a range of efficiencies similar to that of human embryonic stem cells (ESCs). Although three iPSC lines were resistant to neural differentiation, early neuralization rescued their performance. Therefore, all 16 iPSC lines passed a stringent test of differentiation capacity despite variations in karyotype and in the expression of early pluripotency markers and transgenes. This iPSC and ESC test set is a robust resource for those interested in the basic biology of stem cells and their applications.
Figures

Comment in
-
IPSCs put to the test.Nat Biotechnol. 2011 Mar;29(3):233-5. doi: 10.1038/nbt.1805. Nat Biotechnol. 2011. PMID: 21390027 No abstract available.
-
Testing pluripotency.Nat Methods. 2011 Apr;8(4):287. doi: 10.1038/nmeth0411-287. Nat Methods. 2011. PMID: 21574272 No abstract available.
References
-
- Dimos JT, et al. Induced pluripotent stem cells generated from patients with ALS can be differentiated into motor neurons. Science. 2008;321:1218–1221. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

