Evidence for regulatory diversity and auto-regulation at the TAC1 locus in sensory neurones
- PMID: 21294877
- PMCID: PMC3042928
- DOI: 10.1186/1742-2094-8-10
Evidence for regulatory diversity and auto-regulation at the TAC1 locus in sensory neurones
Abstract
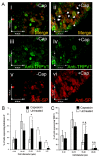
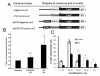
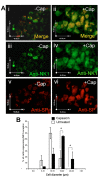
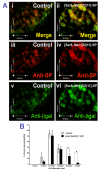
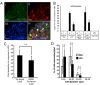
The neuropeptide substance-P (SP) is expressed from the TAC1 gene in sensory neurones where it acts as a key modulator of neurogenic inflammation. The promoter of TAC1 (TAC1prom) plays a central role in the regulation of the TAC1 gene but requires the presence of a second regulatory element; ECR2, to support TAC1 expression in sensory neurones and to respond appropriately to signalling pathways such as MAPkinases and noxious induction by capsaicin. We examined whether the effect of capsaicin on ECR2-TAC1prom activity in larger diameter neurones was cell autonomous or non- cell autonomous. We demonstrate that TRPV1 is not expressed in all the same cells as SP following capsaicin induction suggesting the presence of a non-cell autonomous mechanism for TAC1 up-regulation following capsaicin induction. In addition, we demonstrate that induction of SP and ECR1-TAC1prom activity in these larger diameter neurones can be induced by potassium depolarisation suggesting that, in addition to capsaicin induction, transgene activity may be modulated by voltage gated calcium channels. Furthermore, we show that NK1 is expressed in all SP- expressing cells after capsaicin induction and that an agonist of NK1 can activate both SP and the transgene in larger diameter neurones. These observations suggest the presence of an autocrine loop that controls the expression of the TAC1 promoter in sensory neurones. In contrast, induction of the TAC1 promoter by LPS was not dependent on ECR2 and did not occur in large diameter neurones. These studies demonstrate the diversity of mechanisms modulating the activity of the TAC1 promoter and provide novel directions for the development of new anti-inflammatory therapies.
Figures
Similar articles
-
Long-range regulatory synergy is required to allow control of the TAC1 locus by MEK/ERK signalling in sensory neurones.Neurosignals. 2010;18(3):173-85. doi: 10.1159/000322010. Epub 2010 Dec 16. Neurosignals. 2010. PMID: 21160161 Free PMC article.
-
TRPV1, NK1 receptor and substance P immunoreactivity and gene expression in the rat lumbosacral spinal cord and urinary bladder after systemic, low dose vanilloid administration.Regul Pept. 2011 Apr 11;167(2-3):250-8. doi: 10.1016/j.regpep.2011.02.004. Epub 2011 Feb 15. Regul Pept. 2011. PMID: 21329730
-
Functional effects of polymorphisms on glucocorticoid receptor modulation of human anxiogenic substance-P gene promoter activity in primary amygdala neurones.Psychoneuroendocrinology. 2014 Sep;47(100):43-55. doi: 10.1016/j.psyneuen.2014.04.017. Epub 2014 May 2. Psychoneuroendocrinology. 2014. PMID: 25001955 Free PMC article.
-
TRPV1 and SP: key elements for sepsis outcome?Br J Pharmacol. 2013 Dec;170(7):1279-92. doi: 10.1111/bph.12056. Br J Pharmacol. 2013. PMID: 23145480 Free PMC article. Review.
-
Capsaicin and sensory neurones: a historical perspective.Prog Drug Res. 2014;68:1-37. doi: 10.1007/978-3-0348-0828-6_1. Prog Drug Res. 2014. PMID: 24941663 Review.
Cited by
-
Exploring the effects of polymorphisms on cis-regulatory signal transduction response.Trends Mol Med. 2013 Feb;19(2):99-107. doi: 10.1016/j.molmed.2012.11.003. Epub 2012 Dec 19. Trends Mol Med. 2013. PMID: 23265842 Free PMC article. Review.
-
Transcriptome analysis of the human corneal endothelium.Invest Ophthalmol Vis Sci. 2014 Nov 6;55(12):7821-30. doi: 10.1167/iovs.14-15021. Invest Ophthalmol Vis Sci. 2014. PMID: 25377225 Free PMC article.
-
Topical tissue nanotransfection of Prox1 is effective in the prophylactic management of lymphedema.Mol Ther Nucleic Acids. 2024 Jan 18;35(1):102121. doi: 10.1016/j.omtn.2024.102121. eCollection 2024 Mar 12. Mol Ther Nucleic Acids. 2024. PMID: 38333673 Free PMC article.
-
Differential activity by polymorphic variants of a remote enhancer that supports galanin expression in the hypothalamus and amygdala: implications for obesity, depression and alcoholism.Neuropsychopharmacology. 2011 Oct;36(11):2211-21. doi: 10.1038/npp.2011.93. Epub 2011 Jun 29. Neuropsychopharmacology. 2011. PMID: 21716262 Free PMC article.
-
An analysis of possible off target effects following CAS9/CRISPR targeted deletions of neuropeptide gene enhancers from the mouse genome.Neuropeptides. 2017 Aug;64:101-107. doi: 10.1016/j.npep.2016.11.003. Epub 2016 Nov 4. Neuropeptides. 2017. PMID: 27866656 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

