Sex differences in the contribution of ATP-sensitive K+ channels in trigeminal ganglia under an acute muscle pain condition
- PMID: 21296645
- PMCID: PMC3124308
- DOI: 10.1016/j.neuroscience.2011.01.045
Sex differences in the contribution of ATP-sensitive K+ channels in trigeminal ganglia under an acute muscle pain condition
Abstract
In this study, we examined whether functional subunits of the ATP-dependent K+ channel (KATP) are expressed in trigeminal ganglia (TG), which contains sensory neurons that innervate oral and facial structures. We also investigated whether direct activation of the KATP effectively attenuates mechanical hypersensitivity in the context of an acute orofacial muscle pain condition. The KATP expression in TG and behavioral studies were conducted in age matched male and female Sprague-Dawley rats. RT-PCR experiments showed that the mRNAs for the inwardly rectifying pore-forming subunits, Kir6.1 and Kir6.2, as well as the regulatory sulfonylurea subunits, SUR1 and SUR2, were reliably detected in TG. Subsequent western blot analysis confirmed that proteins for all four subunits are expressed in TG, and showed that Kir6.2 is expressed at a significantly higher level in male TG compared to that of female rats. This observation was confirmed by the immunohistochemical demonstration of higher percentages of Kir6 positive masseter afferents in female rats. Masseteric injection of capsaicin evokes a time dependent increase in masseter sensitivity to noxious mechanical stimulation. A specific KATP agonist, pinacidil, dose-dependently attenuated the capsaicin-induced mechanical hypersensitivity in male rats. The dose of pinacidil (20 μg) that completely blocked the capsaicin responses in male rats was ineffective in female rats regardless of their estrus phases. Only at the highest dose (300 μg) we used, pinacidil was partially effective in female rats. Similarly, another KATP agonist, diazoxide which targets different KATP subunits also showed sex specific responses in attenuating capsaicin-induced masseter hypersensitivity. These data suggested that sex differences in functional KATP expression in TG may underlie sex specific responses to KATP agonists. The present study provided novel information on sex differences in KATP expression in TG and its contribution under an orofacial muscle pain condition.
Copyright © 2011 IBRO. Published by Elsevier Ltd. All rights reserved.
Conflict of interest statement
Figures

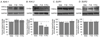




References
-
- Amarante LH, Alves DP, Duarte ID. Study of the involvement of K+ channels in the peripheral antinociception of the kappa-opioid receptor agonist bremazocine. Eur J Pharmacol. 2004;494:155–160. - PubMed
-
- Amoroso S, Schmid-Antomarchi H, Fosset M, Lazdunski M. Glucose, sulfonylureas, and neurotransmitter release: role of ATP-sensitive K+ channels. Science. 1990;247(4944):852–854. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

