Immunolocalization of an alternative respiratory chain in Antonospora (Paranosema) locustae spores: mitosomes retain their role in microsporidial energy metabolism
- PMID: 21296913
- PMCID: PMC3127642
- DOI: 10.1128/EC.00283-10
Immunolocalization of an alternative respiratory chain in Antonospora (Paranosema) locustae spores: mitosomes retain their role in microsporidial energy metabolism
Abstract
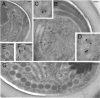
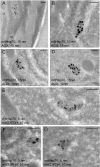
Microsporidia are a group of fungus-related intracellular parasites with severely reduced metabolic machinery. They lack canonical mitochondria, a Krebs cycle, and a respiratory chain but possess genes encoding glycolysis enzymes, a glycerol phosphate shuttle, and ATP/ADP carriers to import host ATP. The recent finding of alternative oxidase genes in two clades suggests that microsporidial mitosomes may retain an alternative respiratory pathway. We expressed the fragments of mitochondrial chaperone Hsp70 (mitHsp70), mitochondrial glycerol-3-phosphate dehydrogenase (mitG3PDH), and alternative oxidase (AOX) from the microsporidium Antonospora (Paranosema) locustae in Escherichia coli. Immunoblotting with antibodies against recombinant polypeptides demonstrated specific accumulation of both metabolic enzymes in A. locustae spores. At the same time comparable amounts of mitochondrial Hsp70 were found in spores and in stages of intracellular development as well. Immunoelectron microscopy of ultrathin cryosections of spores confirmed mitosomal localization of the studied proteins. Small amounts of enzymes of an alternative respiratory chain in merogonial and early sporogonial stages, alongside their accumulation in mature spores, suggest conspicuous changes in components and functions of mitosomes during the life cycle of microsporidia and the important role of these organelles in parasite energy metabolism, at least at the final stages of sporogenesis.
Figures

Similar articles
-
Heterologous expression of pyruvate dehydrogenase E1 subunits of the microsporidium Paranosema (Antonospora) locustae and immunolocalization of the mitochondrial protein in amitochondrial cells.FEMS Microbiol Lett. 2009 Apr;293(2):285-91. doi: 10.1111/j.1574-6968.2009.01545.x. Epub 2009 Feb 26. FEMS Microbiol Lett. 2009. PMID: 19260967
-
Pyruvate-converting activity in the spores of the microsporidian genus Paranosema (Antonospora).FEMS Microbiol Lett. 2006 Jun;259(1):142-6. doi: 10.1111/j.1574-6968.2006.00259.x. FEMS Microbiol Lett. 2006. PMID: 16684114
-
[Immunolocalization of microsporidium paranosema locustae canning lSP70 family proteins in locust infected fat body].Parazitologiia. 2014 Jan-Feb;48(1):63-70. Parazitologiia. 2014. PMID: 25434239 Russian.
-
Back to basics: a revealing secondary reduction of the mitochondrial protein import pathway in diverse intracellular parasites.Biochim Biophys Acta. 2013 Feb;1833(2):295-303. doi: 10.1016/j.bbamcr.2012.02.006. Epub 2012 Feb 16. Biochim Biophys Acta. 2013. PMID: 22366436 Review.
-
Shrink it or lose it: balancing loss of function with shrinking genomes in the microsporidia.Virulence. 2011 Jan-Feb;2(1):67-70. doi: 10.4161/viru.2.1.14606. Epub 2011 Jan 1. Virulence. 2011. PMID: 21217203 Review.
Cited by
-
The genome of the obligate intracellular parasite Trachipleistophora hominis: new insights into microsporidian genome dynamics and reductive evolution.PLoS Pathog. 2012;8(10):e1002979. doi: 10.1371/journal.ppat.1002979. Epub 2012 Oct 25. PLoS Pathog. 2012. PMID: 23133373 Free PMC article.
-
A new family of cell surface located purine transporters in Microsporidia and related fungal endoparasites.Elife. 2019 Jul 29;8:e47037. doi: 10.7554/eLife.47037. Elife. 2019. PMID: 31355745 Free PMC article.
-
Protists in the Insect Rearing Industry: Benign Passengers or Potential Risk?Insects. 2022 May 21;13(5):482. doi: 10.3390/insects13050482. Insects. 2022. PMID: 35621816 Free PMC article. Review.
-
Invasion of Host Cells by Microsporidia.Front Microbiol. 2020 Feb 18;11:172. doi: 10.3389/fmicb.2020.00172. eCollection 2020. Front Microbiol. 2020. PMID: 32132983 Free PMC article. Review.
-
Morphology and Transcriptome Analysis of Nosema bombycis Sporoplasm and Insights into the Initial Infection of Microsporidia.mSphere. 2020 Feb 12;5(1):e00958-19. doi: 10.1128/mSphere.00958-19. mSphere. 2020. PMID: 32051240 Free PMC article.
References
-
- Abrahamsen M. S., et al. 2004. Complete genome sequence of the apicomplexan Cryptosporidium parvum. Science 304:441–445 - PubMed
-
- Beznoussenko G. V., et al. 2007. Analogs of the Golgi complex in microsporidia: structure and avesicular mechanisms of function. J. Cell Sci. 120:1288–1298 - PubMed
-
- Bradford M. 1976. A rapid and sensitive method for the quantitation of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72:248–254 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

