Concussive brain trauma in the mouse results in acute cognitive deficits and sustained impairment of axonal function
- PMID: 21299360
- PMCID: PMC3070143
- DOI: 10.1089/neu.2010.1729
Concussive brain trauma in the mouse results in acute cognitive deficits and sustained impairment of axonal function
Abstract
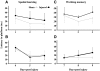
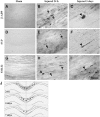
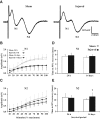
Concussive brain injury (CBI) accounts for approximately 75% of all brain-injured people in the United States each year and is particularly prevalent in contact sports. Concussion is the mildest form of diffuse traumatic brain injury (TBI) and results in transient cognitive dysfunction, the neuropathologic basis for which is traumatic axonal injury (TAI). To evaluate the structural and functional changes associated with concussion-induced cognitive deficits, adult mice were subjected to an impact on the intact skull over the midline suture that resulted in a brief apneic period and loss of the righting reflex. Closed head injury also resulted in an increase in the wet weight:dry weight ratio in the cortex suggestive of edema in the first 24 h, and the appearance of Fluoro-Jade-B-labeled degenerating neurons in the cortex and dentate gyrus of the hippocampus within the first 3 days post-injury. Compared to sham-injured mice, brain-injured mice exhibited significant deficits in spatial acquisition and working memory as measured using the Morris water maze over the first 3 days (p<0.001), but not after the fourth day post-injury. At 1 and 3 days post-injury, intra-axonal accumulation of amyloid precursor protein in the corpus callosum and cingulum was accompanied by neurofilament dephosphorylation, impaired transport of Fluoro-Gold and synaptophysin, and deficits in axonal conductance. Importantly, deficits in retrograde transport and in action potential of myelinated axons continued to be observed until 14 days post-injury, at which time axonal degeneration was apparent. These data suggest that despite recovery from acute cognitive deficits, concussive brain trauma leads to axonal degeneration and a sustained perturbation of axonal function.
Figures








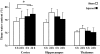


Similar articles
-
Midline brain injury in the immature rat induces sustained cognitive deficits, bihemispheric axonal injury and neurodegeneration.Exp Neurol. 2008 Sep;213(1):84-92. doi: 10.1016/j.expneurol.2008.05.009. Epub 2008 May 23. Exp Neurol. 2008. PMID: 18599043 Free PMC article.
-
Temporal window of vulnerability to repetitive experimental concussive brain injury.Neurosurgery. 2005 Feb;56(2):364-74; discussion 364-74. doi: 10.1227/01.neu.0000149008.73513.44. Neurosurgery. 2005. PMID: 15670384
-
Mild fluid percussion injury in mice produces evolving selective axonal pathology and cognitive deficits relevant to human brain injury.J Neurotrauma. 2010 Aug;27(8):1429-38. doi: 10.1089/neu.2010.1288. J Neurotrauma. 2010. PMID: 20528171
-
Translational Considerations for Behavioral Impairment and Rehabilitation Strategies after Diffuse Traumatic Brain Injury.In: Kobeissy FH, editor. Brain Neurotrauma: Molecular, Neuropsychological, and Rehabilitation Aspects. Boca Raton (FL): CRC Press/Taylor & Francis; 2015. Chapter 36. In: Kobeissy FH, editor. Brain Neurotrauma: Molecular, Neuropsychological, and Rehabilitation Aspects. Boca Raton (FL): CRC Press/Taylor & Francis; 2015. Chapter 36. PMID: 26269926 Free Books & Documents. Review.
-
Development of Concepts in the Pathology of Traumatic Axonal and Traumatic Brain Injury.In: Kobeissy FH, editor. Brain Neurotrauma: Molecular, Neuropsychological, and Rehabilitation Aspects. Boca Raton (FL): CRC Press/Taylor & Francis; 2015. Chapter 3. In: Kobeissy FH, editor. Brain Neurotrauma: Molecular, Neuropsychological, and Rehabilitation Aspects. Boca Raton (FL): CRC Press/Taylor & Francis; 2015. Chapter 3. PMID: 26269901 Free Books & Documents. Review.
Cited by
-
Nigella sativa extract abrogates traumatic brain injury-induced memory impairment in adult mice.Heliyon. 2024 Sep 18;10(18):e38106. doi: 10.1016/j.heliyon.2024.e38106. eCollection 2024 Sep 30. Heliyon. 2024. PMID: 39381232 Free PMC article.
-
Pre-Clinical Traumatic Brain Injury Common Data Elements: Toward a Common Language Across Laboratories.J Neurotrauma. 2015 Nov 15;32(22):1725-35. doi: 10.1089/neu.2014.3861. Epub 2015 Aug 27. J Neurotrauma. 2015. PMID: 26058402 Free PMC article.
-
Characterization of Traumatic Brain Injury in a Gyrencephalic Ferret Model Using the Novel Closed Head Injury Model of Engineered Rotational Acceleration (CHIMERA).Neurotrauma Rep. 2023 Nov 9;4(1):761-780. doi: 10.1089/neur.2023.0047. eCollection 2023. Neurotrauma Rep. 2023. PMID: 38028274 Free PMC article.
-
A Systematic Review of Closed Head Injury Models of Mild Traumatic Brain Injury in Mice and Rats.J Neurotrauma. 2019 Jun;36(11):1683-1706. doi: 10.1089/neu.2018.6127. Epub 2019 Mar 6. J Neurotrauma. 2019. PMID: 30661454 Free PMC article.
-
Repeated mild closed head injury in neonatal rats results in sustained cognitive deficits associated with chronic microglial activation and neurodegeneration.J Neuropathol Exp Neurol. 2023 Jul 20;82(8):707-721. doi: 10.1093/jnen/nlad048. J Neuropathol Exp Neurol. 2023. PMID: 37390808 Free PMC article.
References
-
- Baker A.J. Phan N. Moulton R.J. Fehlings M.G. Yucel Y. Zhao M. Liu E. Tian G.F. Attenuation of the electrophysiological function of the corpus callosum after fluid percussion injury in the rat. J. Neurotrauma. 2002;19:587–599. - PubMed
-
- Bazarian J.J. McClung J. Shah M.N. Cheng Y.T. Flesher W. Kraus J. Mild traumatic brain injury in the United States, 1998–2000. Brain Inj. 2005;19:85–91. - PubMed
-
- Blumbergs P.C. Scott G. Manavis J. Wainwright H. Simpson D.A. McLean A.J. Staining of amyloid precursor protein to study axonal damage in mild head injury. Lancet. 1994;344:1055–1056. - PubMed
-
- Buki A. Povlishock J.T. All roads lead to disconnection?—Traumatic axonal injury revisited. Acta Neurochir. (Wien.) 2006;148:181–193. discussion 193–194. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

