Synthesis, receptor binding, and CNS pharmacological studies of new thyrotropin-releasing hormone (TRH) analogues
- PMID: 21302359
- PMCID: PMC3479646
- DOI: 10.1002/cmdc.201000481
Synthesis, receptor binding, and CNS pharmacological studies of new thyrotropin-releasing hormone (TRH) analogues
Abstract
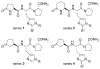
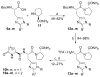
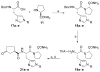
As part of our search for selective and CNS-active thyrotropin-releasing hormone (TRH) analogues, we synthesized a set of 44 new analogues in which His and pGlu residues were modified or replaced. The analogues were evaluated as agonists at TRH-R1 and TRH-R2 in cells in vitro, and in vivo in mice for analeptic and anticonvulsant activities. Several analogues bound to TRH-R1 and TRH-R2 with good to moderate affinities, and are full agonists at both receptor subtypes. Specifically, analogue 21 a (R=CH3) exhibited binding affinities (Ki values) of 0.17 μM for TRH-R1 and 0.016 μM for TRH-R2; it is 10-fold less potent than TRH in binding to TRH-R1 and equipotent with TRH in binding to TRH-R2. Compound 21 a, the most selective agonist, activated TRH-R2 with a potency (EC50 value) of 0.0021 μM, but activated TRH-R1 at EC50=0.05 μM, and exhibited 24-fold selectivity for TRH-R2 over TRH-R1. The newly synthesized TRH analogues were also evaluated in vivo to assess their potencies in antagonism of barbiturate-induced sleeping time, and several analogues displayed potent analeptic activity. Specifically, analogues 21 a,b and 22 a,b decreased sleeping time by nearly 50% more than TRH. These analogues also displayed potent anticonvulsant activity and provided significant protection against PTZ-induced seizures, but failed to provide any protection in MES-induced seizures at 10 μmol kg(-1). The results of this study provide evidence that TRH analogues that show selectivity for TRH-R2 over TRH-R1 possess potent CNS activity.
Copyright © 2011 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
Figures

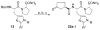
Similar articles
-
Synthesis, receptor binding, and activation studies of N(1)-alkyl-L-histidine containing thyrotropin-releasing hormone (TRH) analogues.Bioorg Med Chem. 2006 Sep 1;14(17):5981-8. doi: 10.1016/j.bmc.2006.05.031. Epub 2006 Jun 2. Bioorg Med Chem. 2006. PMID: 16735122
-
Thyrotropin-releasing hormone (TRH) analogues that exhibit selectivity to TRH receptor subtype 2.J Med Chem. 2005 Sep 22;48(19):6162-5. doi: 10.1021/jm0505462. J Med Chem. 2005. PMID: 16162016
-
Modifications of the pyroglutamic acid and histidine residues in thyrotropin-releasing hormone (TRH) yield analogs with selectivity for TRH receptor type 2 over type 1.Bioorg Med Chem. 2007 Jan 1;15(1):433-43. doi: 10.1016/j.bmc.2006.09.045. Epub 2006 Oct 10. Bioorg Med Chem. 2007. PMID: 17035026
-
Thyrotropin-releasing hormone receptors -- similarities and differences.J Mol Endocrinol. 2003 Apr;30(2):87-97. doi: 10.1677/jme.0.0300087. J Mol Endocrinol. 2003. PMID: 12683933 Review.
-
Chemistry and biology of thyrotropin-releasing hormone (TRH) and its analogs.Curr Med Chem. 2008;15(26):2718-33. doi: 10.2174/092986708786242912. Curr Med Chem. 2008. PMID: 18991632 Review.
Cited by
-
Unmet needs of patients with narcolepsy: perspectives on emerging treatment options.Nat Sci Sleep. 2015 May 22;7:51-61. doi: 10.2147/NSS.S56077. eCollection 2015. Nat Sci Sleep. 2015. PMID: 26045680 Free PMC article. Review.
-
Taltirelin alleviates fatigue-like behavior in mouse models of cancer-related fatigue.Pharmacol Res. 2017 Oct;124:1-8. doi: 10.1016/j.phrs.2017.07.012. Epub 2017 Jul 15. Pharmacol Res. 2017. PMID: 28720519 Free PMC article.
-
Discovery of a low affinity thyrotropin-releasing hormone (TRH)-like peptide that exhibits potent inhibition of scopolamine-induced memory impairment in mice.RSC Adv. 2015 Jun 23;5:56872-56884. doi: 10.1039/c5ra06935a. RSC Adv. 2015. PMID: 26191403 Free PMC article.
-
Hypothalamic neurohormones and immune responses.Front Integr Neurosci. 2013 Aug 13;7:56. doi: 10.3389/fnint.2013.00056. eCollection 2013. Front Integr Neurosci. 2013. PMID: 23964208 Free PMC article.
References
-
- Horita A, Carino MA, Lai H. Ann Rev Pharmacol Toxicol. 1986;26:311–332. - PubMed
-
- Nillni EA, Sevarino KA. Endocr Rev. 1999;20:599–648. - PubMed
-
- Burgus R, Dunn JF, Desiderio D, Guillemin R. C R Acad Sci Hebd Seances Acad Sci D. 1969;269:1870–1873. - PubMed
-
- Boler J, Enzmann F, Folkers K, Bowers CY, Schally AV. Biochem Biophys Res Commun. 1969;37:705–710. - PubMed
-
- Schaner P, Todd RB, Seidah NG, Nillini EA. J Biol Chem. 1997;272:19958–19968. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

