Hypoxia activates the notch signaling pathway in cells of the intervertebral disc: implications in degenerative disc disease
- PMID: 21305512
- PMCID: PMC3613279
- DOI: 10.1002/art.30246
Hypoxia activates the notch signaling pathway in cells of the intervertebral disc: implications in degenerative disc disease
Abstract
Objective: To investigate whether hypoxia regulates Notch signaling, and whether Notch plays a role in intervertebral disc cell proliferation.
Methods: Reverse transcription-polymerase chain reaction and Western blotting were used to measure expression of Notch signaling components in intervertebral disc tissue from mature rats and from human discs. Transfections were performed to determine the effects of hypoxia and Notch on target gene activity.
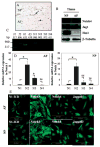
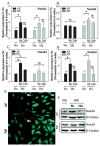
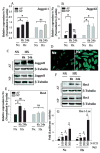
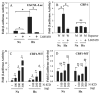
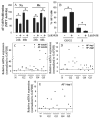
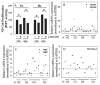
Results: Cells of the nucleus pulposus and annulus fibrosus of rat disc tissue expressed components of the Notch signaling pathway. Expression of Notch-2 was higher than that of the other Notch receptors in both the nucleus pulposus and annulus fibrosus. In both tissues, hypoxia increased Notch1 and Notch4 messenger RNA (mRNA) expression. In the annulus fibrosus, mRNA expression of the Notch ligand Jagged1 was induced by hypoxia, while Jagged2 mRNA expression was highly sensitive to hypoxia in both tissues. A Notch signaling inhibitor, L685458, blocked hypoxic induction of the activity of the Notch-responsive luciferase reporters 12xCSL and CBF1. Expression of the Notch target gene Hes1 was induced by hypoxia, while coexpression with the Notch-intracellular domain increased Hes1 promoter activity. Moreover, inhibition of Notch signaling blocked disc cell proliferation. Analysis of human disc tissue showed that there was increased expression of Notch signaling proteins in degenerated discs.
Conclusion: In intervertebral disc cells, hypoxia promotes expression of Notch signaling proteins. Notch signaling is an important process in the maintenance of disc cell proliferation, and thus offers a therapeutic target for the restoration of cell numbers during degenerative disc disease.
Copyright © 2011 by the American College of Rheumatology.
Figures
Comment in
-
New evidence of the role of the hypoxia-regulated pathway in nucleus pulposus cell survival: comment on the article by Hiyama et al.Arthritis Rheum. 2012 Mar;64(3):940-1; author reply 941-2. doi: 10.1002/art.33480. Arthritis Rheum. 2012. PMID: 22095167 No abstract available.
Similar articles
-
Inflammatory cytokines induce NOTCH signaling in nucleus pulposus cells: implications in intervertebral disc degeneration.J Biol Chem. 2013 Jun 7;288(23):16761-16774. doi: 10.1074/jbc.M112.446633. Epub 2013 Apr 15. J Biol Chem. 2013. PMID: 23589286 Free PMC article. Clinical Trial.
-
Cell type-specific effects of Notch signaling activation on intervertebral discs: Implications for intervertebral disc degeneration.J Cell Physiol. 2018 Jul;233(7):5431-5440. doi: 10.1002/jcp.26385. Epub 2018 Jan 19. J Cell Physiol. 2018. PMID: 30025440 Free PMC article.
-
Transplantation of gene-modified nucleus pulposus cells reverses rabbit intervertebral disc degeneration.Chin Med J (Engl). 2011 Aug;124(16):2431-7. Chin Med J (Engl). 2011. PMID: 21933582
-
Notochordal cells in the adult intervertebral disc: new perspective on an old question.Crit Rev Eukaryot Gene Expr. 2011;21(1):29-41. doi: 10.1615/critreveukargeneexpr.v21.i1.30. Crit Rev Eukaryot Gene Expr. 2011. PMID: 21967331 Free PMC article. Review.
-
Insights into the Notch signaling pathway in degenerative musculoskeletal disorders: Mechanisms and perspectives.Biomed Pharmacother. 2023 Dec 31;169:115884. doi: 10.1016/j.biopha.2023.115884. Epub 2023 Nov 17. Biomed Pharmacother. 2023. PMID: 37981460 Review.
Cited by
-
The E3 ubiquitin ligase SCFFBXL14 complex stimulates neuronal differentiation by targeting the Notch signaling factor HES1 for proteolysis.J Biol Chem. 2017 Dec 8;292(49):20100-20112. doi: 10.1074/jbc.M117.815001. Epub 2017 Oct 25. J Biol Chem. 2017. PMID: 29070679 Free PMC article.
-
Circadian factors BMAL1 and RORα control HIF-1α transcriptional activity in nucleus pulposus cells: implications in maintenance of intervertebral disc health.Oncotarget. 2016 Apr 26;7(17):23056-71. doi: 10.18632/oncotarget.8521. Oncotarget. 2016. PMID: 27049729 Free PMC article.
-
Inflammatory cytokines induce NOTCH signaling in nucleus pulposus cells: implications in intervertebral disc degeneration.J Biol Chem. 2013 Jun 7;288(23):16761-16774. doi: 10.1074/jbc.M112.446633. Epub 2013 Apr 15. J Biol Chem. 2013. PMID: 23589286 Free PMC article. Clinical Trial.
-
Regenerative medicine for the treatment of chronic low back pain: a narrative review.J Int Med Res. 2023 Feb;51(2):3000605231155777. doi: 10.1177/03000605231155777. J Int Med Res. 2023. PMID: 36802994 Free PMC article. Review.
-
Multiscale Regulation of the Intervertebral Disc: Achievements in Experimental, In Silico, and Regenerative Research.Int J Mol Sci. 2021 Jan 12;22(2):703. doi: 10.3390/ijms22020703. Int J Mol Sci. 2021. PMID: 33445782 Free PMC article. Review.
References
-
- Gruber HE, Ashraf N, Kilburn J, Williams C, Norton HJ, Gordon BE, et al. Vertebral endplate architecture and vascularization: application of micro-computerized tomography, a vascular tracer, and immunocytochemistry in analyses of disc degeneration in the aging sand rat. Spine. 2005;30:2593–600. - PubMed
-
- Hassler O. The human intervertebral disc: a micro-angiographical study on its vascular supply at various ages. Acta Orthop Scand. 1969;40:765–72. - PubMed
-
- Rudert M, Tillmann B. Lymph and blood supply of the human intervertebral disc: cadaver study of correlations to discitis. Acta Orthop Scand. 1993;64:37–40. - PubMed
-
- Bartels EM, Fairbank JC, Winlove CP, Urban JP. Oxygen and lactate concentrations measured in vivo in the intervertebral discs of patients with scoliosis and back pain. Spine. 1998;23:1–7. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

