Oral ondansetron versus domperidone for symptomatic treatment of vomiting during acute gastroenteritis in children: multicentre randomized controlled trial
- PMID: 21310051
- PMCID: PMC3045958
- DOI: 10.1186/1471-2431-11-15
Oral ondansetron versus domperidone for symptomatic treatment of vomiting during acute gastroenteritis in children: multicentre randomized controlled trial
Abstract
Background: Vomiting in children with acute gastroenteritis (AG) is not only a direct cause of fluid loss but it is also a major factor of failure of oral rehydration therapy (ORT). Physicians who provide care to paediatric patients in the emergency department (ED) usually prescribe intravenous fluid therapy (IVT) for mild or moderate dehydration when vomiting is the major symptom. Thus, effective symptomatic treatment of vomiting would lead to an important reduction in the use of IVT and, consequently, of the duration of hospital stay and of frequency of hospital admission. Available evidence on symptomatic treatment of vomiting shows the efficacy of the most recently registered molecule (ondansetron) but a proper evaluation of antiemetics drugs largely used in clinical practice, such as domperidone, is lacking.
Objectives: To compare the efficacy of ondansetron and domperidone for the symptomatic treatment of vomiting in children with AG who have failed ORT.
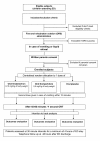
Methods/design: Multicentre, double-blind randomized controlled trial conducted in paediatric EDs. Children aged from 1 to 6 years who vomiting, with a presumptive clinical diagnosis of AG, and without severe dehydration will be included. After the failure of a initial ORS administration in ED, eligible children will be randomized to receive: 1) ondansetron syrup (0,15 mg/Kg of body weight); 2) domperidone syrup (0,5 mg/Kg of body weight); 3) placebo. The main study outcome will be the percentage of patients needing nasogastric or IVT after symptomatic oral treatment failure, defined as vomiting or fluid refusal after a second attempt of ORT. Data relative to study outcomes will be collected at 30 minute intervals for a minimum of 6 hours. A telephone follow up call will be made 48 hours after discharge. A total number of 540 children (i.e. 180 patients in each arm) will be enrolled.
Discussion: The trial results would provide evidence on the efficacy of domperidone, which is largely used in clinical practice despite the lack of proper evaluation and a controversial safety profile, as compared to ondansetron, which is not yet authorized in Italy despite evidence supporting its efficacy in treating vomiting. The trial results would contribute to a reduction in the use of IVT and, consequently, in hospital admissions in children with AG. The design of this RCT, which closely reflect current clinical practice in EDs, will allow immediate transferability of results.
Trial registration: ClinicalTrials.gov: NCT01257672.
References
-
- Fontana M, Federico P, Cantucci S. Il ricovero pediatrico in Lombardia: cause, variabilità e possibile inappropriatezza. Medico e Bambino. 2007;26:520–6.
-
- King CK, Glass R, Bresee JS, Duggan C. Managing acute gastroenteritis among children: oral rehydration, maintenance, and nutritional therapy. MMWR Recomm Rep. 2003;52(RR-16):1–16. - PubMed
-
- Guarino A, Albano F, Ashkenazi S. et al.European Society for Paediatric Gastroenterology, Hepatology, and Nutrition/European Society for Paediatric Infectious Diseases evidence-based guidelines for the management of acute gastroenteritis in children in Europe: executive summary. J Pediatr Gastroenterol Nutr. 2008;46:619–621. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous