Assessment of epicutaneous testing of a monovalent Influenza A (H1N1) 2009 vaccine in egg allergic patients
- PMID: 21314925
- PMCID: PMC3045365
- DOI: 10.1186/1710-1492-7-3
Assessment of epicutaneous testing of a monovalent Influenza A (H1N1) 2009 vaccine in egg allergic patients
Abstract
Background: H1N1 is responsible for the first influenza pandemic in 41 years. In the fall of 2009, an H1N1 vaccine became available in Canada with the hopes of reducing the overall effect of the pandemic. The purpose of this study was to assess the safety of administering 2 different doses of a monovalent split virus 2009 H1N1 vaccine in egg allergic patients.
Methods: Patients were skin tested to the H1N1 vaccine in the outpatient paediatric and adult allergy and immunology clinics of the Health Sciences Centre and Children's Hospital of Winnipeg, Manitoba Canada. Individuals <9 years of age were administered 1.88 μg's of hem-agglutinin antigen per 0.25 ml dose and individuals ≥9 years were administered 3.75 μg's of hemagglutinin antigen per 0.5 ml dose. Upon determination of a negative skin test, the vaccine was administered with a 30 minute observation period.
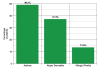
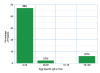
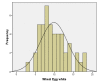
Results: A total of 61 patients with egg allergy (history of an allergic reaction to egg with either positive skin test &/or specific IgE to egg >0.35 Ku/L) were referred to our allergy clinics for skin testing to the H1N1 vaccine. 2 patients were excluded, one did not have a skin prick test to the H1N1 vaccine (only vaccine administration) and the other passed an egg challenge during the study period. Ages ranged from 1 to 27 years (mean 5.6 years). There were 41(69.5%) males and 18(30.5%) females. All but one patient with a history of egg allergy, positive skin test to egg and/or elevated specific IgE level to egg had negative skin tests to the H1N1 vaccine. The 58 patients with negative skin testing to the H1N1 vaccine were administered the vaccine and observed for 30 minutes post vaccination with no adverse results. The patient with the positive skin test to the H1N1 vaccine was also administered the vaccine intramuscularly with no adverse results.
Conclusions: Despite concern regarding possible anaphylaxis to the H1N1 vaccine in egg allergic patients, in our case series 1/59(1.7%) patients with sensitization to egg were also sensitized to the H1N1 vaccine. Administration of the H1N1 vaccine in egg allergic patients with negative H1N1 skin tests and observation is safe. Administering the vaccine in a 1 or 2 dose protocol without skin testing is a reasonable alternative as per the CSACI guidelines.
Figures
References
-
- The Canadian Society of Allergy and Clinical Immunology. Statement: administration of H1N1 and seasonal influenza vaccine to egg allergic individuals. http://www.csaci.ca/include/files/CSACI_H1N1_Statement.pdf Accessed July 23, 2010.
LinkOut - more resources
Full Text Sources