An unrestrained proinflammatory M1 macrophage population induced by iron impairs wound healing in humans and mice
- PMID: 21317534
- PMCID: PMC3049372
- DOI: 10.1172/JCI44490
An unrestrained proinflammatory M1 macrophage population induced by iron impairs wound healing in humans and mice
Abstract
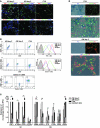
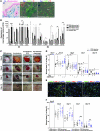
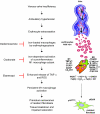
Uncontrolled macrophage activation is now considered to be a critical event in the pathogenesis of chronic inflammatory diseases such as atherosclerosis, multiple sclerosis, and chronic venous leg ulcers. However, it is still unclear which environmental cues induce persistent activation of macrophages in vivo and how macrophage-derived effector molecules maintain chronic inflammation and affect resident fibroblasts essential for tissue homeostasis and repair. We used a complementary approach studying human subjects with chronic venous leg ulcers, a model disease for macrophage-driven chronic inflammation, while establishing a mouse model closely reflecting its pathogenesis. Here, we have shown that iron overloading of macrophages--as was found to occur in human chronic venous leg ulcers and the mouse model--induced a macrophage population in situ with an unrestrained proinflammatory M1 activation state. Via enhanced TNF-α and hydroxyl radical release, this macrophage population perpetuated inflammation and induced a p16(INK4a)-dependent senescence program in resident fibroblasts, eventually leading to impaired wound healing. This study provides insight into the role of what we believe to be a previously undescribed iron-induced macrophage population in vivo. Targeting this population may hold promise for the development of novel therapies for chronic inflammatory diseases such as chronic venous leg ulcers.
Figures



Comment in
-
Macrophages: Iron macrophages.Nat Rev Immunol. 2011 Mar;11(3):158. doi: 10.1038/nri2947. Nat Rev Immunol. 2011. PMID: 21452590 No abstract available.
Similar articles
-
Iron and iron-dependent reactive oxygen species in the regulation of macrophages and fibroblasts in non-healing chronic wounds.Free Radic Biol Med. 2019 Mar;133:262-275. doi: 10.1016/j.freeradbiomed.2018.09.036. Epub 2018 Sep 24. Free Radic Biol Med. 2019. PMID: 30261274 Review.
-
Role of microRNA-21 and Its Underlying Mechanisms in Inflammatory Responses in Diabetic Wounds.Int J Mol Sci. 2020 May 8;21(9):3328. doi: 10.3390/ijms21093328. Int J Mol Sci. 2020. PMID: 32397166 Free PMC article.
-
The proinflammatory cytokines IL-1β and TNF-α modulate corneal epithelial wound healing through p16Ink4a suppressing STAT3 activity.J Cell Physiol. 2020 Dec;235(12):10081-10093. doi: 10.1002/jcp.29823. Epub 2020 May 31. J Cell Physiol. 2020. PMID: 32474927
-
Iron overloaded polarizes macrophage to proinflammation phenotype through ROS/acetyl-p53 pathway.Cancer Med. 2018 Aug;7(8):4012-4022. doi: 10.1002/cam4.1670. Epub 2018 Jul 10. Cancer Med. 2018. PMID: 29989329 Free PMC article.
-
Oxidative stress in chronic venous leg ulcers.Wound Repair Regen. 2005 Sep-Oct;13(5):452-61. doi: 10.1111/j.1067-1927.2005.00065.x. Wound Repair Regen. 2005. PMID: 16176453 Review.
Cited by
-
Macrophage Ablation Reduces M2-Like Populations and Jeopardizes Tumor Growth in a MAFIA-Based Glioma Model.Neoplasia. 2015 Apr;17(4):374-84. doi: 10.1016/j.neo.2015.03.003. Neoplasia. 2015. PMID: 25925380 Free PMC article.
-
Functional muscle recovery with nanoparticle-directed M2 macrophage polarization in mice.Proc Natl Acad Sci U S A. 2018 Oct 16;115(42):10648-10653. doi: 10.1073/pnas.1806908115. Epub 2018 Oct 1. Proc Natl Acad Sci U S A. 2018. PMID: 30275293 Free PMC article.
-
Human umbilical cord mesenchymal stem cells alleviate ongoing autoimmune dacryoadenitis in rabbits via polarizing macrophages into an anti-inflammatory phenotype.Exp Eye Res. 2020 Feb;191:107905. doi: 10.1016/j.exer.2019.107905. Epub 2019 Dec 28. Exp Eye Res. 2020. PMID: 31891674 Free PMC article.
-
Energy-Supporting Enzyme-Mimic Nanoscaffold Facilitates Tendon Regeneration Based on a Mitochondrial Protection and Microenvironment Remodeling Strategy.Adv Sci (Weinh). 2022 Nov;9(31):e2202542. doi: 10.1002/advs.202202542. Epub 2022 Aug 24. Adv Sci (Weinh). 2022. PMID: 36000796 Free PMC article.
-
MSCs rescue impaired wound healing in a murine LAD1 model by adaptive responses to low TGF-β1 levels.EMBO Rep. 2020 Apr 3;21(4):e49115. doi: 10.15252/embr.201949115. Epub 2020 Feb 21. EMBO Rep. 2020. PMID: 32080965 Free PMC article.
References
-
- Agren MS, et al. Causes and effects of the chronic inflammation in venous leg ulcers. Acta Derm Venereol Suppl (Stockh). 2000;210:3–17. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

