High dose atorvastatin decreases cellular markers of immune activation without affecting HIV-1 RNA levels: results of a double-blind randomized placebo controlled clinical trial
- PMID: 21325137
- PMCID: PMC3071124
- DOI: 10.1093/infdis/jiq115
High dose atorvastatin decreases cellular markers of immune activation without affecting HIV-1 RNA levels: results of a double-blind randomized placebo controlled clinical trial
Abstract
Background: 3-hydroxy-3-methyl-glutaryl-coenzyme A reductase inhibitors (statins) exhibit antiviral activity against human immunodeficiency virus type 1 (HIV-1) in vitro and may modulate the immune response to HIV infection. Studies evaluating the antiviral activity of statins have yielded conflicting results.
Methods: We conducted a randomized, double-blind, placebo-controlled crossover trial to investigate the effect of atorvastatin on HIV-1 RNA (primary objective) and cellular markers of immune activation (secondary objective). HIV-infected individuals not receiving antiretroviral therapy were randomized to receive either 8 weeks of atorvastatin (80 mg) or placebo daily. After a 4-6 week washout phase, participants switched treatment assignments. The study had 80% power to detect a 0.3 log(10) decrease in HIV-1 RNA level. Expression of CD38 and HLA-DR on CD4(+) and CD8(+) T cells was used to measure immune activation.
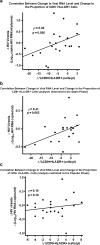
Results: Of 24 randomized participants, 22 completed the study. Although HIV-1 RNA level was unaffected by the intervention (-0.13 log(10) copies/mL; P = .85), atorvastatin use resulted in reductions in circulating proportions of CD4(+) HLA-DR(+) (-2.5%; P = .02), CD8(+) HLA-DR(+) (-5%; P = .006), and CD8(+) HLA-DR(+) CD38(+) T cells (-3%; P = .03). Reductions in immune activation did not correlate with declines in serum levels of low-density lipoprotein cholesterol.
Conclusions: Short-term use of atorvastatin was associated with modest but statistically significant reductions in the proportion of activated T lymphocytes.
Figures


Comment in
-
Statins as anti-inflammatory therapy in HIV disease?J Infect Dis. 2011 Mar 15;203(6):751-2. doi: 10.1093/infdis/jiq120. Epub 2011 Jan 25. J Infect Dis. 2011. PMID: 21266514 Free PMC article. No abstract available.
-
Reply to Ganesan et al.J Infect Dis. 2012 Feb 1;205(3):518-9. doi: 10.1093/infdis/jir758. Epub 2011 Nov 28. J Infect Dis. 2012. PMID: 22124133 No abstract available.
References
-
- Deeks SG. Immune dysfunction, inflammation, and accelerated aging in patients on antiretroviral therapy. Top HIV Med. 2009;17:118–23. - PubMed
-
- Friis-Moller N, Reiss P, Sabin CA, et al. Class of antiretroviral drugs and the risk of myocardial infarction. N Engl J Med. 2007;356:1723–35. - PubMed
-
- Pike LJ. Rafts defined: a report on the Keystone Symposium on Lipid Rafts and Cell Function. J Lipid Res. 2006;47:1597–8. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

