The autism risk genes MET and PLAUR differentially impact cortical development
- PMID: 21328570
- PMCID: PMC3644181
- DOI: 10.1002/aur.172
The autism risk genes MET and PLAUR differentially impact cortical development
Abstract
Candidate risk genes for autism spectrum disorder (ASD) have been identified, but the challenge of determining their contribution to pathogenesis remains. We previously identified two ASD risk genes encoding the receptor tyrosine kinase MET and the urokinase plasminogen activator receptor (PLAUR), which is thought to modulate availability of the MET ligand. We also reported a role for Met signaling in cortical interneuron development in vitro and a reduction of these neurons in uPAR (mouse ortholog of PLAUR) null mice, suggesting that disruption of either gene impacts cortical development similarly. Here, we modify this conclusion, reporting that interneuron numbers are unchanged in the neocortex of Met(fx/fx) / Dlx5/6(cre) mice, in which Met is ablated from cells arising from the ventral telencephalon (VTel). Consistent with this, Met transcript is not detected in the VTel during interneuron genesis and migration; furthermore, during the postnatal period of interneuron maturation, Met is co-expressed in glutamatergic projection neurons, but not interneurons. Low levels of Met protein are expressed in the VTel at E12.5 and E14.5, likely reflecting the arrival of Met containing corticofugal axons. Met expression, however, is induced in E12.5 VTel cells after 2 days in vitro, perhaps underlying discrepancies between observations in vitro and in Met(fx/fx) / Dlx5/6(cre) mice. We suggest that, in vivo, Met impacts the development of cortical projection neurons, whereas uPAR influences interneuron maturation. An altered balance between excitation and inhibition has been postulated as a biological mechanism for ASD; this imbalance could arise from different risk genes differentially affecting either or both elements.
Copyright © 2010, International Society for Autism Research, Wiley Periodicals, Inc.
Figures
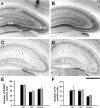

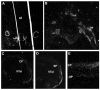
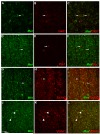
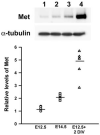
Comment in
-
Commentary for special issue of Autism research on mouse models in ASD: a clinical perspective.Autism Res. 2011 Feb;4(1):1-4. doi: 10.1002/aur.185. Autism Res. 2011. PMID: 21328567 No abstract available.
References
-
- Abercrombie M. Estimation of nuclear population from microtome sections. Anatomical Record. 1946;94:239–247. - PubMed
-
- Anderson SA, Eisenstat DD, Shi L, Rubenstein JL. Interneuron migration from basal forebrain to neocortex: dependence on Dlx genes. Science. 1997;278:474–476. - PubMed
-
- Anderson SA, Marin O, Horn C, Jennings K, Rubenstein JL. Distinct cortical migrations from the medial and lateral ganglionic eminences. Development. 2001;128:353–363. - PubMed
-
- Bahi A, Boyer F, Kafri T, Dreyer JL. Silencing urokinase in the ventral tegmental area in vivo induces changes in cocaine-induced hyperlocomotion. Journal of Neurochemistry. 2006;98:1619–1631. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

