Mitochondrial peptidase IMMP2L mutation causes early onset of age-associated disorders and impairs adult stem cell self-renewal
- PMID: 21332923
- PMCID: PMC3111879
- DOI: 10.1111/j.1474-9726.2011.00686.x
Mitochondrial peptidase IMMP2L mutation causes early onset of age-associated disorders and impairs adult stem cell self-renewal
Abstract
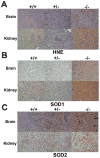
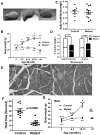
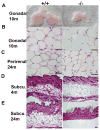
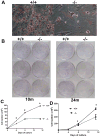
Mitochondrial reactive oxygen species (ROS) are proposed to play a central role in aging and age-associated disorders, although direct in vivo evidence is lacking. We recently generated a mouse mutant with mutated inner mitochondrial membrane peptidase 2-like (Immp2l) gene, which impairs the signal peptide sequence processing of mitochondrial proteins cytochrome c1 and glycerol phosphate dehydrogenase 2. The mitochondria from mutant mice generate elevated levels of superoxide ion and cause impaired fertility in both sexes. Here, we design experiments to examine the effects of excessive mitochondrial ROS generation on health span. We show that Immp2l mutation increases oxidative stress in multiple organs such as the brain and the kidney, although expression of superoxide dismutases in these tissues of the mutants is also increased. The mutants show multiple aging-associated phenotypes, including wasting, sarcopenia, loss of subcutaneous fat, kyphosis, and ataxia, with female mutants showing earlier onset and more severe age-associated disorders than male mutants. The loss of body weight and fat was unrelated to food intake. Adipose-derived stromal cells (ADSC) from mutant mice showed impaired proliferation capability, formed significantly less and smaller colonies in colony formation assays, although they retained adipogenic differentiation capability in vitro. This functional impairment was accompanied by increased levels of oxidative stress. Our data showed that mitochondrial ROS is the driving force of accelerated aging and suggested that ROS damage to adult stem cells could be one of the mechanisms for age-associated disorders.
© 2011 The Authors. Aging Cell © 2011 Blackwell Publishing Ltd/Anatomical Society of Great Britain and Ireland.
Figures

References
-
- Behrens M, Michaelis G, Pratje E. Mitochondrial inner membrane protease 1 of Saccharomyces cerevisiae shows sequence similarity to the Escherichia coli leader peptidase. Mol Gen Genet. 1991;228:167–176. - PubMed
-
- Caplan AI. Adult mesenchymal stem cells for tissue engineering versus regenerative medicine. Journal of Cellular Physiology. 2007;213:341–347. - PubMed
-
- Chehab O, Ouertani M, Souiden Y, Chaieb K, Mahdouani K. Plasma antioxidants and human aging: a study on healthy elderly Tunisian population. Mol Biotechnol. 2008;40:27–37. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

