Outcomes of patients with advanced non-small cell lung cancer treated in a phase I clinic
- PMID: 21339262
- PMCID: PMC3228100
- DOI: 10.1634/theoncologist.2010-0308
Outcomes of patients with advanced non-small cell lung cancer treated in a phase I clinic
Abstract
Background: The outcomes of patients with advanced non-small cell lung cancer (NSCLC) treated in phase I clinical trials have not been systematically analyzed.
Methods: We reviewed the records of consecutive patients with advanced/metastatic NSCLC who were treated in the Phase I Clinical Trials Program at MD Anderson from August 2004 to May 2009.
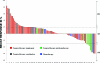
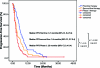
Results: Eighty-five patients (51 men, 34 women) treated on various phase I protocols were identified. The median age was 62 years (range, 30-85). The median number of previous systemic therapies was two (range, 0-5). A partial response was observed in eight patients (9.5%) and stable disease lasting >4 months was observed in 16 patients (19%). The median overall survival time was 10.6 months and median progression-free survival (PFS) time was 2.8 months, which was 0.6 months shorter than the median PFS of 3.4 months following prior second-line therapy. Factors predicting longer survival in the univariate analysis were an Eastern Cooperative Oncology Group performance status (PS) score of 0-1, no prior smoking, two or fewer organ systems involved, a hemoglobin level ≥ 12 g/dL, liver metastases, a history of thromboembolism, and a platelets count > 440 × 10(9)/L. In the multivariate analysis, a PS score of 0-1 and history negative for smoking predicted longer survival. Sixty-two (73%) patients had grade ≤ 2 toxicity, and there were no treatment-related deaths.
Conclusion: Phase I clinical trials were well tolerated by selected patients with advanced NSCLC treated at M.D. Anderson. Nonsmokers and patients with a good PS survived longer. PFS in our population was shorter in smokers/ex-smokers and patients with a PS score of 2. It is reasonable to refer pretreated patients with a good PS to phase I clinical trials.
Conflict of interest statement
The content of this article has been reviewed by independent peer reviewers to ensure that it is balanced, objective, and free from commercial bias. No financial relationships relevant to the content of this article have been disclosed by the authors or independent peer reviewers.
Figures

References
-
- National Cancer Institute. SEER Cancer Statistics Review 1975–2006. Estimated New Cancer Cases and Deaths for 2009. [accessed November 29, 2010]. Available at http://www.seer.cancer.gov/csr/1975_2006/results_single/sect_01_table.01....
-
- Shepherd FA, Dancey J, Ramlau R, et al. Prospective randomized trial of docetaxel versus best supportive care in patients with non-small-cell lung cancer previously treated with platinum-based chemotherapy. J Clin Oncol. 2000;18:2095–2103. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

