Signaling thresholds govern heterogeneity in IL-7-receptor-mediated responses of naïve CD8(+) T cells
- PMID: 21339767
- PMCID: PMC3342499
- DOI: 10.1038/icb.2011.5
Signaling thresholds govern heterogeneity in IL-7-receptor-mediated responses of naïve CD8(+) T cells
Abstract
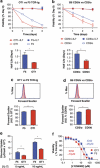
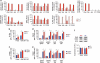
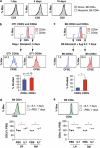
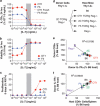
Variable sensitivity to T-cell-receptor (TCR)- and IL-7-receptor (IL-7R)-mediated homeostatic signals among naïve T cells has thus far been largely attributed to differences in TCR specificity. We show here that even when withdrawn from self-peptide-induced TCR stimulation, CD8(+) T cells exhibit heterogeneous responses to interleukin-7 (IL-7) that are mechanistically associated with IL-7R expression differences that correlate with relative CD5 expression. Whereas CD5(hi) and CD5(lo) T cells survive equivalently in the presence of saturating IL-7 levels in vitro, CD5(hi) T cells proliferate more robustly. Conversely, CD5(lo) T cells exhibit prolonged survival when withdrawn from homeostatic stimuli. Through quantitative experimental analysis of signaling downstream of IL-7R, we find that the enhanced IL-7 responsiveness of CD5(hi) T cells is directly related to their greater surface IL-7R expression. Further, we identify a quantitative threshold in IL-7R-mediated signaling capacity required for proliferation that lies well above an analogous threshold requirement for survival. These distinct thresholds allow subtle differences in IL-7R expression between CD5(lo) and CD5(hi) T cells to give rise to significant variations in their respective IL-7-induced proliferation, without altering survival. Heterogeneous IL-7 responsiveness is observed similarly in vivo, with CD5(hi) naïve T cells proliferating preferentially in lymphopenic mice or lymphoreplete mice administered with exogenous IL-7. However, IL-7 in lymphoreplete mice appears to be maintained at an effective level for preserving homeostasis, such that neither CD5(hi) IL-7R(hi) nor CD5(lo) IL-7R(lo) T cells proliferate or survive preferentially. Our findings indicate that IL-7R-mediated signaling not only maintains the size but also impacts the diversity of the naïve T-cell repertoire.
Figures




References
-
- Takada K, Jameson SC. Naive T cell homeostasis: from awareness of space to a sense of place. Nat Rev Immunol. 2009;9:823–832. - PubMed
-
- Surh CD, Sprent J. Homeostasis of naive and memory T cells. Immunity. 2008;29:848–862. - PubMed
-
- Mahajan VS, Leskov IB, Chen JZ. Homeostasis of T cell diversity. Cell Mol Immunol. 2005;2:1–10. - PubMed
-
- Geiselhart LA, Humphries CA, Gregorio TA, Mou S, Subleski J, Komschlies KL. IL-7 administration alters the CD4:CD8 ratio, increases T cell numbers, and increases T cell function in the absence of activation. J Immunol. 2001;166:3019–3027. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

