RNA interference targeting slug increases cholangiocarcinoma cell sensitivity to cisplatin via upregulating PUMA
- PMID: 21339993
- PMCID: PMC3039959
- DOI: 10.3390/ijms12010385
RNA interference targeting slug increases cholangiocarcinoma cell sensitivity to cisplatin via upregulating PUMA
Retraction in
-
Retraction: Zhang, K., et al. Knockdown of Snail Sensitizes Pancreatic Cancer Cells to Chemotherapeutic Agents and Irradiation. Int. J. Mol. Sci. 2010, 11, 4891-4904, doi:10.3390/ijms11124891 and Zhang, K., et al. RNA Interference Targeting Slug Increases Cholangiocarcinoma Cell Sensitivity to Cisplatin via Upregulating PUMA. Int. J. Mol. Sci. 2011, 12, 385-400, doi:10.3390/ijms12010385.Int J Mol Sci. 2017 Jul 10;18(7):1483. doi: 10.3390/ijms18071483. Int J Mol Sci. 2017. PMID: 28698505 Free PMC article. No abstract available.
Abstract
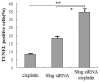
Slug is an E-cadherin repressor and a suppressor of PUMA (p53 upregulated modulator of apoptosis) and it has recently been demonstrated that Slug plays an important role in controlling apoptosis. In this study, we examined whether Slug's ability to silence expression suppresses the growth of cholangiocarcinoma cells and/or sensitizes cholangiocarcinoma cells to chemotherapeutic agents through induction of apoptosis. We targeted the Slug gene using siRNA (Slug siRNA) via full Slug cDNA plasmid (Slug cDNA) transfection of cholangiocarcinoma cells. Slug siRNA, cisplatin, or Slug siRNA in combination with cisplatin, were used to treat cholangiocarcinoma cells in vitro. Western blot was used to detect the expression of Slug, PUMA, and E-cadherin protein. TUNEL, Annexin V Staining, and cell cycle analysis were used to detect apoptosis. A nude mice subcutaneous xenograft model of QBC939 cells was used to assess the effect of Slug silencing and/or cisplatin on tumor growth. Immunohistochemical staining was used to analyze the expression of Slug and PUMA. TUNEL was used to detect apoptosis in vivo. The results showed that PUMA and E-cadherin expression in cholangiocarcinoma cells is Slug dependent. We demonstrated that Slug silencing and cisplatin both promote apoptosis by upregulation of PUMA, not by upregulation of E-cadherin. Slug silencing significantly sensitized cholangiocarcinoma cells to cisplatin through upregulation of PUMA. Finally, we showed that Slug silencing suppressed the growth of QBC939 xenograft tumors and sensitized the tumor cells to cisplatin through PUMA upregulation and induction of apoptosis. Our findings indicate that Slug is an important modulator of the therapeutic response of cholangiocarcinoma cells and is potentially useful as a sensitizer in cholangiocarcinoma therapy. One of the mechanisms is the regulation of PUMA by Slug.
Keywords: E-cadherin; PUMA; chemotherapy; cisplatin; slug.
Figures





Similar articles
-
Slug inhibition upregulates radiation-induced PUMA activity leading to apoptosis in cholangiocarcinomas.Med Oncol. 2011 Dec;28 Suppl 1:S301-9. doi: 10.1007/s12032-010-9759-x. Epub 2010 Dec 1. Med Oncol. 2011. PMID: 21120639
-
Small interfering RNA (siRNA) against Slug induces apoptosis and sensitizes human anaplastic thyroid carcinoma cells to doxorubicin.Cancer Biomark. 2017;18(4):357-366. doi: 10.3233/CBM-160192. Cancer Biomark. 2017. PMID: 28128736
-
Slug regulates proliferation and invasiveness of esophageal adenocarcinoma cells in vitro and in vivo.Med Oncol. 2011 Dec;28(4):1089-100. doi: 10.1007/s12032-010-9652-7. Epub 2010 Aug 21. Med Oncol. 2011. PMID: 20730573
-
Decreased expression of MUC1 induces apoptosis and inhibits migration in pancreatic cancer PANC-1 cells via regulation of Slug pathway.Cancer Biomark. 2017 Dec 6;20(4):469-476. doi: 10.3233/CBM-170297. Cancer Biomark. 2017. PMID: 28869438
-
Targeting SLUG sensitizes leukemia cells to ADR-induced apoptosis.Int J Clin Exp Med. 2015 Dec 15;8(12):22139-48. eCollection 2015. Int J Clin Exp Med. 2015. PMID: 26885188 Free PMC article.
Cited by
-
Defining the molecular signature of chemotherapy-mediated lung tumor phenotype modulation and increased susceptibility to T-cell killing.Cancer Biother Radiopharm. 2012 Feb;27(1):23-35. doi: 10.1089/cbr.2012.1203. Epub 2012 Feb 8. Cancer Biother Radiopharm. 2012. PMID: 22316209 Free PMC article.
-
SLUG is activated by nuclear factor kappa B and confers human alveolar epithelial A549 cells resistance to tumor necrosis factor-alpha-induced apoptosis.World J Surg Oncol. 2013 Jan 22;11:12. doi: 10.1186/1477-7819-11-12. World J Surg Oncol. 2013. PMID: 23339680 Free PMC article.
-
PUMA Cooperates with p21 to Regulate Mammary Epithelial Morphogenesis and Epithelial-To-Mesenchymal Transition.PLoS One. 2013 Jun 21;8(6):e66464. doi: 10.1371/journal.pone.0066464. Print 2013. PLoS One. 2013. PMID: 23805223 Free PMC article.
-
Knockdown of Akt sensitizes osteosarcoma cells to apoptosis induced by cisplatin treatment.Int J Mol Sci. 2011;12(5):2994-3005. doi: 10.3390/ijms12052994. Epub 2011 May 10. Int J Mol Sci. 2011. PMID: 21686164 Free PMC article.
-
Liver carcinogenesis: rodent models of hepatocarcinoma and cholangiocarcinoma.Dig Liver Dis. 2013 Jun;45(6):450-9. doi: 10.1016/j.dld.2012.10.008. Epub 2012 Nov 22. Dig Liver Dis. 2013. PMID: 23177172 Free PMC article. Review.
References
-
- De Groen PC, Gores GJ, LaRusso NF, Gunderson LL, Nagorney DM. Biliary tract cancers. N. Engl. J. Med. 1999;341:1368–1378. - PubMed
-
- Malouf G, Dreyer C, Guedj N, Paradis V, Degos F, Belghiti J, Le Tourneau C, Faivre S, Raymond E. Prognosis factors of cholangiocarcinoma: contribution of recent molecular biology tools. Bull Cancer. 2009;96:405–415. - PubMed
-
- Wehbe H, Henson R, Lang M, Meng F, Patel T. Pifithrin-alpha enhances chemosensitivity by a p38 mitogen-activated protein kinase-dependent modulation of the eukaryotic initiation factor 4E in malignant cholangiocytes. J. Pharmacol. Exp. Ther. 2006;319:1153–1161. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

