New ultrahigh affinity host-guest complexes of cucurbit[7]uril with bicyclo[2.2.2]octane and adamantane guests: thermodynamic analysis and evaluation of M2 affinity calculations
- PMID: 21341773
- PMCID: PMC3065999
- DOI: 10.1021/ja109904u
New ultrahigh affinity host-guest complexes of cucurbit[7]uril with bicyclo[2.2.2]octane and adamantane guests: thermodynamic analysis and evaluation of M2 affinity calculations
Abstract
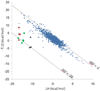
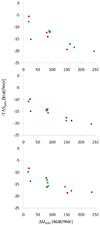
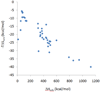
A dicationic ferrocene derivative has previously been shown to bind cucurbit[7]uril (CB[7]) in water with ultrahigh affinity (ΔG(o) = -21 kcal/mol). Here, we describe new compounds that bind aqueous CB[7] equally well, validating our prior suggestion that they, too, would be ultrahigh affinity CB[7] guests. The present guests, which are based upon either a bicyclo[2.2.2]octane or adamantane core, have no metal atoms, so these results also confirm that the remarkably high affinities of the ferrocene-based guest need not be attributed to metal-specific interactions. Because we used the M2 method to compute the affinities of several of the new host-guest systems prior to synthesizing them, the present results also provide for the first blinded evaluation of this computational method. The blinded calculations agree reasonably well with experiment and successfully reproduce the observation that the new adamantane-based guests achieve extremely high affinities, despite the fact that they position a cationic substituent at only one electronegative portal of the CB[7] host. However, there are also significant deviations from experiment, and these lead to the correction of a procedural error and an instructive evaluation of the sensitivity of the calculations to physically reasonable variations in molecular energy parameters. The new experimental and computational results presented here bear on the physical mechanisms of molecular recognition, the accuracy of the M2 method, and the usefulness of host-guest systems as test-beds for computational methods.
Figures







References
-
- Freeman W, Mock W, Shih N. J. Am. Chem. Soc. 1981;103:7367–7368.
-
- Kim J, Jung I, Kim S, Lee E, Kang J, Sakamoto S, Yamaguchi K, Kim K. J. Am. Chem. Soc. 2000;122:540–541.
-
- Liu S, Ruspic C, Mukhopadhyay P, Chakrabarti S, Zavalij PY, Isaacs L. J. Am. Chem. Soc. 2005;127:15959–15967. - PubMed
-
- Chang C, Gilson MK. J. Am. Chem. Soc. 2004;126:13156–13164. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

