Structure-activity relationship of capsaicin analogs and transient receptor potential vanilloid 1-mediated human lung epithelial cell toxicity
- PMID: 21343315
- PMCID: PMC3083109
- DOI: 10.1124/jpet.110.178491
Structure-activity relationship of capsaicin analogs and transient receptor potential vanilloid 1-mediated human lung epithelial cell toxicity
Abstract
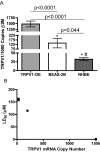
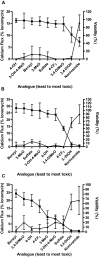
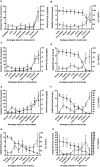
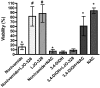
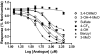
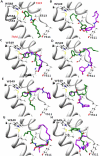
Activation of intracellular transient receptor potential vanilloid-1 (TRPV1) in human lung cells causes endoplasmic reticulum (ER) stress, increased expression of proapoptotic GADD153 (growth arrest- and DNA damage-inducible transcript 3), and cytotoxicity. However, in cells with low TRPV1 expression, cell death is not inhibited by TRPV1 antagonists, despite preventing GADD153 induction. In this study, chemical variants of the capsaicin analog nonivamide were synthesized and used to probe the relationship between TRPV1 receptor binding, ER calcium release, GADD153 expression, and cell death in TRPV1-overexpressing BEAS-2B, normal BEAS-2B, and primary normal human bronchial epithelial lung cells. Modification of the 3-methoxy-4-hydroxybenzylamide vanilloid ring pharmacophore of nonivamide reduced the potency of the analogs and rendered several analogs mildly inhibitory. Correlation analysis of analog-induced calcium flux, GADD153 induction, and cytotoxicity revealed a direct relationship for all three endpoints in all three lung cell types for nonivamide and N-(3,4-dihydroxybenzyl)nonanamide. However, the N-(3,4-dihydroxybenzyl)nonanamide analog also produced cytotoxicity through redox cycling/reactive oxygen species formation, shown by inhibition of cell death by N-acetylcysteine. Molecular modeling of binding interactions between the analogs and TRPV1 agreed with data for reduced potency of the analogs, and only nonivamide was predicted to form a "productive" ligand-receptor complex. This study provides vital information on the molecular interactions of capsaicinoids with TRPV1 and substantiates TRPV1-mediated ER stress as a conserved mechanism of lung cell death by prototypical TRPV1 agonists.
Figures
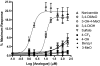
Similar articles
-
Contributions of TRPV1, endovanilloids, and endoplasmic reticulum stress in lung cell death in vitro and lung injury.Am J Physiol Lung Cell Mol Physiol. 2012 Jan 1;302(1):L111-9. doi: 10.1152/ajplung.00231.2011. Epub 2011 Sep 23. Am J Physiol Lung Cell Mol Physiol. 2012. PMID: 21949157 Free PMC article.
-
Transient receptor potential vanilloid 1 agonists cause endoplasmic reticulum stress and cell death in human lung cells.J Pharmacol Exp Ther. 2007 Jun;321(3):830-8. doi: 10.1124/jpet.107.119412. Epub 2007 Mar 1. J Pharmacol Exp Ther. 2007. PMID: 17332266
-
TRPV1 antagonists elevate cell surface populations of receptor protein and exacerbate TRPV1-mediated toxicities in human lung epithelial cells.Toxicol Sci. 2006 Jan;89(1):278-86. doi: 10.1093/toxsci/kfi292. Epub 2005 Aug 24. Toxicol Sci. 2006. PMID: 16120755 Free PMC article.
-
Capsaicinoids cause inflammation and epithelial cell death through activation of vanilloid receptors.Toxicol Sci. 2003 May;73(1):170-81. doi: 10.1093/toxsci/kfg044. Toxicol Sci. 2003. PMID: 12721390 Free PMC article.
-
Nonivamide, a capsaicin analog, increases dopamine and serotonin release in SH-SY5Y cells via a TRPV1-independent pathway.Mol Nutr Food Res. 2013 Nov;57(11):2008-18. doi: 10.1002/mnfr.201200846. Epub 2013 Aug 9. Mol Nutr Food Res. 2013. PMID: 23929722
Cited by
-
Contributions of TRPV1, endovanilloids, and endoplasmic reticulum stress in lung cell death in vitro and lung injury.Am J Physiol Lung Cell Mol Physiol. 2012 Jan 1;302(1):L111-9. doi: 10.1152/ajplung.00231.2011. Epub 2011 Sep 23. Am J Physiol Lung Cell Mol Physiol. 2012. PMID: 21949157 Free PMC article.
-
Phytochemicals for the Management of Melanoma.Mini Rev Med Chem. 2016;16(12):953-79. doi: 10.2174/1389557516666160211120157. Mini Rev Med Chem. 2016. PMID: 26864554 Free PMC article. Review.
-
Investigation of anti-microbial and cytotoxic potential of Streptomyces werraensis GRS9 derived from the sediments of river Ganga.Braz J Microbiol. 2025 Jun;56(2):863-882. doi: 10.1007/s42770-025-01642-9. Epub 2025 Mar 11. Braz J Microbiol. 2025. PMID: 40064810
-
Structural mechanism underlying capsaicin binding and activation of the TRPV1 ion channel.Nat Chem Biol. 2015 Jul;11(7):518-524. doi: 10.1038/nchembio.1835. Epub 2015 Jun 8. Nat Chem Biol. 2015. PMID: 26053297 Free PMC article.
-
Non-pungent long chain capsaicin-analogs arvanil and olvanil display better anti-invasive activity than capsaicin in human small cell lung cancers.Cell Adh Migr. 2017 Jan 2;11(1):80-97. doi: 10.1080/19336918.2016.1187368. Epub 2016 May 19. Cell Adh Migr. 2017. PMID: 27196129 Free PMC article.
References
-
- Caterina MJ, Schumacher MA, Tominaga M, Rosen TA, Levine JD, Julius D. (1997) The capsaicin receptor: a heat-activated ion channel in the pain pathway. Nature 389:816–824 - PubMed
-
- Copple IM, Goldring CE, Kitteringham NR, Park BK. (2010) The keap1-nrf2 cellular defense pathway: mechanisms of regulation and role in protection against drug-induced toxicity. Handb Exp Pharmacol 233–266 - PubMed
-
- Gavva NR, Klionsky L, Qu Y, Shi L, Tamir R, Edenson S, Zhang TJ, Viswanadhan VN, Toth A, Pearce LV, et al. (2004) Molecular determinants of vanilloid sensitivity in TRPV1. J Biol Chem 279:20283–20295 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

