"Same day" ex-vivo regional gene therapy: a novel strategy to enhance bone repair
- PMID: 21343916
- PMCID: PMC3098640
- DOI: 10.1038/mt.2011.2
"Same day" ex-vivo regional gene therapy: a novel strategy to enhance bone repair
Abstract
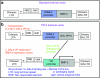
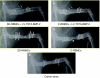
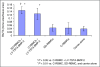
Ex-vivo regional gene therapy with bone marrow cells (BMCs) overexpressing bone morphogenetic protein-2 (BMP-2) has demonstrated efficacy in healing critical sized bone defects in preclinical studies. The purpose of this preclinical study was to compare the osteoinductive potential of a novel "same day" ex-vivo regional gene therapy versus a traditional two-step approach, which involves culture expansion of the donor cells before implantation. In the "same day" strategy buffy coat cells were harvested from the rat bone marrow, transduced with a lentiviral vector-expressing BMP-2 for 1 hour and implanted into a rat femoral defect in the same sitting. There was no significant difference (P = 0.22) with respect to the radiographic healing rates between the femoral defects treated with the "same day" strategy (13/13; 100%) versus the traditional two-step approach (11/14; 78%). However, the femoral defects treated with the "same day" strategy induced earlier radiographic bone healing (P = 0.004) and higher bone volume (BV) [micro-computed tomography (micro-CT); P < 0.001]. The "same day" strategy represents a significant advance in the field of ex-vivo regional gene therapy because it offers a solution to limitations associated with the culture expansion process required in the traditional ex vivo approach. This strategy should be cost-effective when adapted for human use.
Figures



References
-
- Gamradt SC., and, Lieberman JR. Bone graft for revision hip arthroplasty: biology and future applications. Clin Orthop Relat Res: 2003. pp. 183–194. - PubMed
-
- St John TA, Vaccaro AR, Sah AP, Schaefer M, Berta SC, Albert T.et al. (2003Physical and monetary costs associated with autogenous bone graft harvesting Am J Orthop (Belle Mead NJ) 3218–23. - PubMed
-
- Kim DH, Rhim R, Li L, Martha J, Swaim BH, Banco RJ.et al. (2009Prospective study of iliac crest bone graft harvest site pain and morbidity Spine J 9886–892. - PubMed
-
- Govender S, Csimma C, Genant HK, Valentin-Opran A, Amit Y, Arbel R.et al. (2002Recombinant human bone morphogenetic protein-2 for treatment of open tibial fractures: a prospective, controlled, randomized study of four hundred and fifty patients J Bone Joint Surg Am 84-A2123–2134. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

