Interventional MRI-guided putaminal delivery of AAV2-GDNF for a planned clinical trial in Parkinson's disease
- PMID: 21343917
- PMCID: PMC3129792
- DOI: 10.1038/mt.2011.11
Interventional MRI-guided putaminal delivery of AAV2-GDNF for a planned clinical trial in Parkinson's disease
Abstract
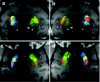
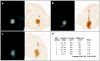
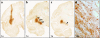
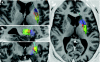
Clinical trials involving direct infusion of neurotrophic therapies for Parkinson's disease (PD) have suffered from poor coverage of the putamen. The planned use of a novel interventional-magnetic resonance imaging (iMRI) targeting system for achieving precise, real-time convection-enhanced delivery in a planned clinical trial of adeno-associated virus serotype 2 (AAV2)-glial-derived neurotrophic factor (GDNF) in PD patients was modeled in nonhuman primates (NHP). NHP received bilateral coinfusions of gadoteridol (Gd)/AAV2-GDNF into two sites in each putamen, and three NHP received larger infusion volumes in the thalamus. The average targeting error for cannula tip placement in the putamen was <1 mm, and adjacent putamenal infusions were distributed in a uniform manner. GDNF expression patterns in the putamen were highly correlated with areas of Gd distribution seen on MRI. The distribution volume to infusion volume ratio in the putamen was similar to that in the thalamus, where larger infusions were achieved. Modeling the placement of adjacent 150 and 300 µl thalamic infusions into the three-dimensional space of the human putamen demonstrated coverage of the postcommissural putamen, containment within the striatum and expected anterograde transport to globus pallidus and substantia nigra pars reticulata. The results elucidate the necessary parameters for achieving widespread GDNF expression in the putamenal motor area and afferent substantia nigra of PD patients.
Figures


References
-
- Love S, Plaha P, Patel NK, Hotton GR, Brooks DJ., and, Gill SS. Glial cell line-derived neurotrophic factor induces neuronal sprouting in human brain. Nat Med. 2005;11:703–704. - PubMed
-
- Ai Y, Markesbery W, Zhang Z, Grondin R, Elseberry D, Gerhardt GA.et al. (2003Intraputamenal infusion of GDNF in aged rhesus monkeys: distribution and dopaminergic effects J Comp Neurol 461250–261. - PubMed
-
- Nutt JG, Burchiel KJ, Comella CL, Jankovic J, Lang AE, Laws ER., Jret al. (2003Randomized, double-blind trial of glial cell line-derived neurotrophic factor (GDNF) in PD Neurology 6069–73. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

