The Bs20x22 anti-CD20-CD22 bispecific antibody has more lymphomacidal activity than do the parent antibodies alone
- PMID: 21347809
- PMCID: PMC3098984
- DOI: 10.1007/s00262-011-0978-6
The Bs20x22 anti-CD20-CD22 bispecific antibody has more lymphomacidal activity than do the parent antibodies alone
Abstract
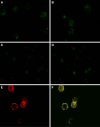
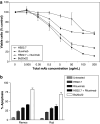
Previous studies have shown that bispecific antibodies that target both CD20 and CD22 have in vivo lymphomacidal properties. We developed a CD20-CD22 bispecific antibody (Bs20x22) from anti-CD20 and the anti-CD22 monoclonal antibodies (mAb), rituximab and HB22.7, respectively. Bs20x22 was constructed using standard methods and was shown to specifically bind CD20 and CD22. In vitro cytotoxicity assays showed that Bs20x22 was three times more effective than either parent mAb alone and twice as effective as a combination of both parent mAb used at equimolar concentrations. Bs20x22 was also nearly four times more effective at inducing apoptosis than either mAb alone. Examination of the MAPK and SAPK signaling cascades revealed that Bs20x22 induced significantly more p38 phosphorylation than either mAb alone. In an in vivo human NHL xenograft model, treatment with Bs20x22 resulted in significantly greater tumor shrinkage and improved overall survival when compared to either mAb alone or treatment with a combination of HB22.7 and rituximab. The effect of the initial tumor volume was assessed by comparing the efficacy of Bs20x22 administered before xenografts grew versus treatment of established tumors; significantly, greater efficacy was found when treatment was initiated before tumors could become established.
Figures


References
-
- Ries L, Melbert D, Krapcho M, Stinchcomb D, Howlader N, Horner M, Mariotto A, Miller B, Feuer E, Altekruse S, Lewis D, Clegg L, Eisner M, Reichman M, Edwards BE. SEER cancer statistics review, 1975–2005. Bethesda, MD: National Cancer Institute; 2008.
-
- Lindley C. The lymphomas: Hodgkin’s disease and non-Hodgkin’s lymphomas. Am Pharm NS. 1991;31:46–51. - PubMed
-
- Schumer ST, Joyce RM (2003) Radioimmunotherapy for Non-Hodgkin’s Lymphoma. Progress Oncol 46–72
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

