Protein dynamics in cytochrome P450 molecular recognition and substrate specificity using 2D IR vibrational echo spectroscopy
- PMID: 21348488
- PMCID: PMC3063108
- DOI: 10.1021/ja109168h
Protein dynamics in cytochrome P450 molecular recognition and substrate specificity using 2D IR vibrational echo spectroscopy
Abstract
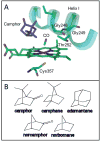
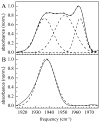
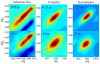
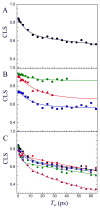
Cytochrome (cyt) P450s hydroxylate a variety of substrates that can differ widely in their chemical structure. The importance of these enzymes in drug metabolism and other biological processes has motivated the study of the factors that enable their activity on diverse classes of molecules. Protein dynamics have been implicated in cyt P450 substrate specificity. Here, 2D IR vibrational echo spectroscopy is employed to measure the dynamics of cyt P450(cam) from Pseudomonas putida on fast time scales using CO bound at the active site as a vibrational probe. The substrate-free enzyme and the enzyme bound to both its natural substrate, camphor, and a series of related substrates are investigated to explicate the role of dynamics in molecular recognition in cyt P450(cam) and to delineate how the motions may contribute to hydroxylation specificity. In substrate-free cyt P450(cam), three conformational states are populated, and the structural fluctuations within a conformational state are relatively slow. Substrate binding selectively stabilizes one conformational state, and the dynamics become faster. Correlations in the observed dynamics with the specificity of hydroxylation of the substrates, the binding affinity, and the substrates' molecular volume suggest that motions on the hundreds of picosecond time scale contribute to the variation in activity of cyt P450(cam) toward different substrates.
Figures
Similar articles
-
Protein dynamics studied with ultrafast two-dimensional infrared vibrational echo spectroscopy.Acc Chem Res. 2012 Nov 20;45(11):1866-74. doi: 10.1021/ar200275k. Epub 2012 Mar 20. Acc Chem Res. 2012. PMID: 22433178 Free PMC article.
-
Role of protein and substrate dynamics in catalysis by Pseudomonas putida cytochrome P450cam.Biochemistry. 2002 Dec 10;41(49):14499-508. doi: 10.1021/bi026379e. Biochemistry. 2002. PMID: 12463748
-
Dynamics underlying hydroxylation selectivity of cytochrome P450cam.Biophys J. 2021 Mar 2;120(5):912-923. doi: 10.1016/j.bpj.2021.01.027. Epub 2021 Feb 3. Biophys J. 2021. PMID: 33545101 Free PMC article.
-
Differential behavior of the sub-sites of cytochrome 450 active site in binding of substrates, and products (implications for coupling/uncoupling).Biochim Biophys Acta. 2007 Mar;1770(3):360-75. doi: 10.1016/j.bbagen.2006.09.018. Epub 2006 Oct 5. Biochim Biophys Acta. 2007. PMID: 17134838 Review.
-
Fourier transform infrared spectroscopy as a tool to study structural properties of cytochromes P450 (CYPs).Anal Bioanal Chem. 2008 Nov;392(6):1031-58. doi: 10.1007/s00216-008-2216-4. Epub 2008 Jun 27. Anal Bioanal Chem. 2008. PMID: 18581103 Review.
Cited by
-
Size-dependent ultrafast structural dynamics inside phospholipid vesicle bilayers measured with 2D IR vibrational echoes.Proc Natl Acad Sci U S A. 2014 Jan 21;111(3):918-23. doi: 10.1073/pnas.1323110111. Epub 2014 Jan 6. Proc Natl Acad Sci U S A. 2014. PMID: 24395796 Free PMC article.
-
Comparison of intrinsic dynamics of cytochrome p450 proteins using normal mode analysis.Protein Sci. 2015 Sep;24(9):1495-507. doi: 10.1002/pro.2737. Epub 2015 Jul 16. Protein Sci. 2015. PMID: 26130403 Free PMC article.
-
Oscillatory Enzyme Dynamics Revealed by Two-Dimensional Infrared Spectroscopy.J Phys Chem Lett. 2016 Jul 7;7(13):2507-11. doi: 10.1021/acs.jpclett.6b01154. Epub 2016 Jun 21. J Phys Chem Lett. 2016. PMID: 27305279 Free PMC article.
-
Anisotropic dynamics of an interfacial enzyme active site observed using tethered substrate analogs and ultrafast 2D IR spectroscopy.J Chem Phys. 2023 Oct 28;159(16):165101. doi: 10.1063/5.0167991. J Chem Phys. 2023. PMID: 37870142 Free PMC article.
-
Applications of two-dimensional infrared spectroscopy.Analyst. 2015 Jul 7;140(13):4336-49. doi: 10.1039/c5an00558b. Analyst. 2015. PMID: 26007625 Free PMC article. Review.
References
-
- Skopalík J, Anzenbacher P, Otyepka M. J Phys Chem B. 2008;112:8165. - PubMed
-
- Wade RC, Motiejunas D, Schleinkofer K, Sudarko, Winn PJ, Banerjee A, Kariakin A, Jung C. Biochim Biophys Acta. 2005;1754:239. - PubMed
-
- Guengerich FP. Chem Res Toxicol. 2008;21:70. - PubMed
-
- Guengerich FP. In: Cytochrome P450: Structure, Mechanism, and Biochemistry. de Montellano O, editor. Vol. 3. Plenum Press; New York: 2005. p. 377.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

