Do two intravenous iron sucrose preparations have the same efficacy?
- PMID: 21355067
- PMCID: PMC3193183
- DOI: 10.1093/ndt/gfr024
Do two intravenous iron sucrose preparations have the same efficacy?
Abstract
Background: Intravenous (i.v.) iron sucrose similar (ISS) preparations are available but clinical comparisons with the originator iron sucrose (IS) are lacking.
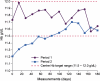
Methods: The impact of switching from IS to ISS on anaemia and iron parameters was assessed in a sequential observational study comparing two periods of 27 weeks each in 75 stable haemodialysis (HD) patients receiving i.v. iron weekly and an i.v. erythropoiesis-stimulating agent (ESA) once every 2 weeks. Patients received IS in the first period (P1) and ISS in the second period (P2).
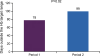
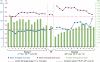
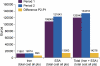
Results: Mean haemoglobin value was 11.78 ± 0.99 g/dL during P1 and 11.48 ± 0.98 g/dL during P2 (P = 0.01). Mean serum ferritin was similar for both treatment periods (P1, 534 ± 328 μg/L; P2, 495 ± 280 μg/L, P = 0.25) but mean TSAT during P1 (49.3 ± 10.9%) was significantly higher than during P2 (24.5 ± 9.4%, P <0.0001). The mean dose of i.v. iron per patient per week was 45.58 ± 32.55 mg in P1 and 61.36 ± 30.98 mg in P2 (+34.6%), while the mean ESA dose was 0.58 ± 0.52 and 0.66 ± 0.64 μg/kg/week, respectively (+13.8%). Total mean anaemia drug costs increased in P2 by 11.9% compared to P1.
Conclusions: The switch from the originator IS to an ISS preparation led to destabilization of a well-controlled population of HD patients and incurred an increase in total anaemia drug costs. Prospective comparative clinical studies are required to prove that ISS are as efficacious and safe as the originator i.v. IS.
Figures
References
-
- Valderrábano F, Hörl WH, Macdougall IC, et al. PRE-dialysis survey on anaemia management. Nephrol Dial Transplant. 2003;18:89–100. - PubMed
-
- Macdougall IC. Iron supplementation in the non-dialysis chronic kidney disease (ND-CKD) patient: oral or intravenous? Curr Med Res Opin. 2010;26:473–482. - PubMed
-
- Locatelli F, Aljama P, Barany P, et al. Revised European best practice guidelines for the management of anaemia in patients with chronic renal failure. Nephrol Dial Transplant. 2004;19(Suppl 2):1–47. - PubMed
-
- National Collaborating Centre for Chronic Conditions. Anaemia Management in Chronic Kidney Disease: National Clinical Guideline for Management in Adults and Children. London: Royal College of Physicians; 2006. pp. 1–172. - PubMed
-
- National Kidney Foundation-Kidney Disease Outcomes Quality Initiative (NKF-KDOQI) KDOQI clinical practice guideline and clinical practice recommendations for anemia in chronic kidney disease: 2007 update of hemoglobin target. Am J Kidney Dis. 2007;50:471–530. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

