Safety profile and clinical activity of multiple subcutaneous doses of MEDI-528, a humanized anti-interleukin-9 monoclonal antibody, in two randomized phase 2a studies in subjects with asthma
- PMID: 21356110
- PMCID: PMC3058114
- DOI: 10.1186/1471-2466-11-14
Safety profile and clinical activity of multiple subcutaneous doses of MEDI-528, a humanized anti-interleukin-9 monoclonal antibody, in two randomized phase 2a studies in subjects with asthma
Abstract
Background: Interleukin-9 (IL-9)-targeted therapies may offer a novel approach for treating asthmatics. Two randomized placebo-controlled studies were conducted to assess the safety profile and potential efficacy of multiple subcutaneous doses of MEDI-528, a humanized anti-IL-9 monoclonal antibody, in asthmatics.
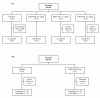
Methods: Study 1: adults (18-65 years) with mild asthma received MEDI-528 (0.3, 1, 3 mg/kg) or placebo subcutaneously twice weekly for 4 weeks. Study 2: adults (18-50 years) with stable, mild to moderate asthma and exercise-induced bronchoconstriction received 50 mg MEDI-528 or placebo subcutaneously twice weekly for 4 weeks. Adverse events (AEs), pharmacokinetics (PK), immunogenicity, asthma control (including asthma exacerbations), and exercise challenge test were evaluated in study 1, study 2, or both.
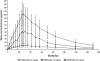
Results: In study 1 (N = 36), MEDI-528 showed linear serum PK; no anti-MEDI-528 antibodies were detected. Asthma control: 1/27 MEDI-528-treated subjects had 1 asthma exacerbation, and 2/9 placebo-treated subjects had a total of 4 asthma exacerbations (one considered a serious AE). In study 2, MEDI-528 (n = 7) elicited a trend in the reduction in mean maximum decrease in FEV1 post-exercise compared to placebo (n = 2) (-6.49% MEDI-528 vs -12.60% placebo; -1.40% vs -20.10%; -5.04% vs -15.20% at study days 28, 56, and 150, respectively). Study 2 was halted prematurely due to a serious AE in an asymptomatic MEDI-528-treated subject who had an abnormal brain magnetic resonance imaging that was found to be an artifact on further evaluation.
Conclusions: In these studies, MEDI-528 showed an acceptable safety profile and findings suggestive of clinical activity that support continued study in subjects with mild to moderate asthma.
Trial registration: ClinicalTrials.gov NCT00507130 NCT00590720.
Figures

References
-
- Current asthma prevalence percents by age, United States: National health interview survey, 2006. 2008. http://www.cdc.gov/asthma/nhis/06/data.htm
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

