Clinical and histopathologic features of fluoroquinolone-induced liver injury
- PMID: 21356330
- PMCID: PMC3718017
- DOI: 10.1016/j.cgh.2011.02.019
Clinical and histopathologic features of fluoroquinolone-induced liver injury
Abstract
Background & aims: Fluoroquinolone-induced liver injury is rare; no prospective studies of well-characterized case series have been published. We studied patients with fluoroquinolone-induced hepatotoxicity from the Drug-Induced Liver Injury Network (DILIN) to characterize injury patterns, outcomes, and associated features.
Methods: We identified subjects with fluoroquinolone hepatotoxicity enrolled in the DILIN from September 2004 to January 2010. Demographic, clinical, and laboratory data were analyzed by descriptive statistical methods.
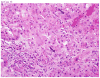
Results: Of the 679 registrants in the DILIN prospective study, 12 had fluoroquinolone hepatotoxicity (6 ciprofloxacin, 4 moxifloxacin, 1 levofloxacin, and 1 gatifloxacin). Seven were women; median age was 57 years (range, 23-80 years), and median time from fluoroquinolone start to symptoms was only 4 days (range, 1-39 days). Nine patients developed symptoms on medication; 3 did so 2, 8, and 32 days after stopping the medication. Cases were equally distributed among hepatocellular injury (predominantly increased levels of alanine aminotransferase), cholestatic injury (predominantly increased levels of alkaline phosphatase), and both. Seven patients had immunoallergic features. Patients with mixed hepatocellular and cholestatic injury had mild disease without jaundice; all recovered. In contrast, 2 of 4 patients with hepatocellular injury and jaundice died, 1 of acute liver failure. One patient with cholestatic injury developed vanishing bile duct syndrome and required liver transplantation; another had a persistently increased serum level of alkaline phosphatase.
Conclusions: Fluoroquinolone liver injury is rapid in onset and often has immunoallergic features, indicating a hypersensitivity reaction. The pattern of injury can be hepatocellular, cholestatic, or mixed; mixed cases are the least severe. Acute and chronic liver failure can occur.
Copyright © 2011 AGA Institute. Published by Elsevier Inc. All rights reserved.
Figures






References
-
- Wolfson JS, Hooper DC. Overview of fluoroquinolone safety. Am J Med. 1991;91(6):153S–161S. - PubMed
-
- Bertino J, Fish D. The safety profile of the fluoroquinolones. Clin Ther. 2000;22(7):798–817. discussion 797; 798–817; discussion 797. - PubMed
-
- Van Bambeke F, Tulkens PM. Safety profile of the respiratory fluoroquinolone moxifloxacin: comparison with other fluoroquinolones and other antibacterial classes. Drug Saf. 2009;32(5):359–78. - PubMed
-
- Kahn JB. Latest industry information on the safety profile of levofloxacin in the US. Chemotherapy. 2001;47(Suppl 3):32–37. discussion 44–48; 32–37; discussion 44–48. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- U01 DK065211/DK/NIDDK NIH HHS/United States
- 2U01-DK065176-06/DK/NIDDK NIH HHS/United States
- 2U01-DK065201-06/DK/NIDDK NIH HHS/United States
- U01 DK083020/DK/NIDDK NIH HHS/United States
- U01 DK065238/DK/NIDDK NIH HHS/United States
- U01 DK065176/DK/NIDDK NIH HHS/United States
- U01 DK083023/DK/NIDDK NIH HHS/United States
- UL1 RR025747/RR/NCRR NIH HHS/United States
- 1U01-DK083027-01/DK/NIDDK NIH HHS/United States
- 5U01-DK065238-08/DK/NIDDK NIH HHS/United States
- UL1 RR025761/RR/NCRR NIH HHS/United States
- 5U01DK065193-04/DK/NIDDK NIH HHS/United States
- U01 DK083027/DK/NIDDK NIH HHS/United States
- U01 DK065201/DK/NIDDK NIH HHS/United States
- 1U01-DK082992-01/DK/NIDDK NIH HHS/United States
- U01 DK065193/DK/NIDDK NIH HHS/United States
- 2U01-DK065184-06/DK/NIDDK NIH HHS/United States
- UL1 RR024982/RR/NCRR NIH HHS/United States
- 2U01-DK065211-06/DK/NIDDK NIH HHS/United States
- 1U01-DK083020-01/DK/NIDDK NIH HHS/United States
- ImNIH/Intramural NIH HHS/United States
- 1U01-DK083023-01/DK/NIDDK NIH HHS/United States
- UL1 RR024150/RR/NCRR NIH HHS/United States
- UL1 RR024986/RR/NCRR NIH HHS/United States
- U01 DK065184/DK/NIDDK NIH HHS/United States
- UL1 RR024134/RR/NCRR NIH HHS/United States
- U01 DK082992/DK/NIDDK NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

