Population size drives industrial Saccharomyces cerevisiae alcoholic fermentation and is under genetic control
- PMID: 21357433
- PMCID: PMC3126379
- DOI: 10.1128/AEM.02547-10
Population size drives industrial Saccharomyces cerevisiae alcoholic fermentation and is under genetic control
Abstract
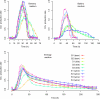
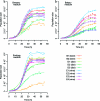
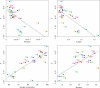
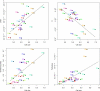
Alcoholic fermentation (AF) conducted by Saccharomyces cerevisiae has been exploited for millennia in three important human food processes: beer and wine production and bread leavening. Most of the efforts to understand and improve AF have been made separately for each process, with strains that are supposedly well adapted. In this work, we propose a first comparison of yeast AFs in three synthetic media mimicking the dough/wort/grape must found in baking, brewing, and wine making. The fermentative behaviors of nine food-processing strains were evaluated in these media, at the cellular, populational, and biotechnological levels. A large variation in the measured traits was observed, with medium effects usually being greater than the strain effects. The results suggest that human selection targeted the ability to complete fermentation for wine strains and trehalose content for beer strains. Apart from these features, the food origin of the strains did not significantly affect AF, suggesting that an improvement program for a specific food processing industry could exploit the variability of strains used in other industries. Glucose utilization was analyzed, revealing plastic but also genetic variation in fermentation products and indicating that artificial selection could be used to modify the production of glycerol, acetate, etc. The major result was that the overall maximum CO(2) production rate (V(max)) was not related to the maximum CO(2) production rate per cell. Instead, a highly significant correlation between V(max) and the maximum population size was observed in all three media, indicating that human selection targeted the efficiency of cellular reproduction rather than metabolic efficiency. This result opens the way to new strategies for yeast improvement.
Figures



References
-
- Albertin W., et al. 2009. Evidence for autotetraploidy associated with reproductive isolation in Saccharomyces cerevisiae: towards a new domesticated species. J. Evol. Biol. 22:2157–2170 - PubMed
-
- Attfield P. V. 1997. Stress tolerance: the key to effective strains of industrial baker's yeast. Nat. Biotechnol. 15:1351–1357 - PubMed
-
- Bely M., Rinaldi A., Dubourdieu D. 2003. Influence of assimilable nitrogen on volatile acidity production by Saccharomyces cerevisiae during high sugar fermentation. J. Biosci. Bioeng. 96:507–512 - PubMed
-
- Bely M., Sablayrolles J. M., Barre P. 1990. Automatic detection of assimilable nitrogen deficiencies during alcoholic fermentation in enological conditions. J. Ferment. Bioeng. 70:246–252
-
- Bely M., Sablayrolles J. M., Barre P. 1990. Description of alcoholic fermentation kinetics—its variability and significance. Am. J. Enol. Vitic. 41:319–324
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

