Potentiation of apoptosis by histone deacetylase inhibitors and doxorubicin combination: cytoplasmic cathepsin B as a mediator of apoptosis in multiple myeloma
- PMID: 21364585
- PMCID: PMC3065279
- DOI: 10.1038/bjc.2011.42
Potentiation of apoptosis by histone deacetylase inhibitors and doxorubicin combination: cytoplasmic cathepsin B as a mediator of apoptosis in multiple myeloma
Abstract
Background: Although inhibitors of histone deacetylase inhibitors (HDACis) in combination with genotoxins potentiate apoptosis, the role of proteases other than caspases in this process remained elusive. Therefore, we examined the potentiation of apoptosis and related mechanisms of HDACis and doxorubicin combination in a panel of myeloma cell lines and in 25 primary myelomas.
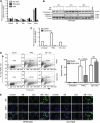
Results: At IC(50) concentrations, sodium butyrate (an HDACi) or doxorubicin alone caused little apoptosis. However, their combination potentiated apoptosis and synergistically reduced the viability of myeloma cells independent of p53 and caspase 3-7 activation. Potentiated apoptosis correlated with nuclear translocation of apoptosis-inducing factor, suggesting the induction of caspase 3- and 7-independent pathways. Consistent with this, butyrate and doxorubicin combination significantly increased the activity of cytoplasmic cathepsin B. Inhibition of cathepsin B either with a small-molecule inhibitor or downregulation with a siRNA reversed butyrate- and doxorubicin-potentiated apoptosis. Finally, ex vivo, clinically relevant concentrations of butyrate or SAHA (suberoylanilide hydroxamic acid, vorinostat, an HDACi in clinical testing) in combination with doxorubicin significantly (P<0.0001) reduced the survival of primary myeloma cells.
Conclusions: Cathepsin B has a prominent function in mediating apoptosis potentiated by HDACi and doxorubicin combinations in myeloma. Our results support a molecular model of lysosomal-mitochondrial crosstalk in HDACi- and doxorubicin-potentiated apoptosis through the activation of cathepsin B.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Badros A, Burger AM, Philip S, Niesvizky R, Kolla SS, Goloubeva O, Harris C, Zwiebel J, Wright JJ, Espinoza-Delgado I, Baer MR, Holleran JL, Egorin MJ, Grant S (2009) Phase I study of vorinostat in combination with bortezomib for relapsed and refractory multiple myeloma. Clin Cancer Res 15(16): 5250–5257 - PMC - PubMed
-
- Bae SI, Cheriyath V, Jacobs BS, Reu FJ, Borden EC (2008) Reversal of methylation silencing of Apo2L/TRAIL receptor 1 (DR4) expression overcomes resistance of SK-MEL-3 and SK-MEL-28 melanoma cells to interferons (IFNs) or Apo2L/TRAIL. Oncogene 27(4): 490–498 - PubMed
-
- Bergsagel PL, Kuehl WM (2001) Chromosome translocations in multiple myeloma. Oncogene 20(40): 5611–5622 - PubMed
-
- Bidere N, Lorenzo HK, Carmona S, Laforge M, Harper F, Dumont C, Senik A (2003) Cathepsin D triggers Bax activation, resulting in selective apoptosis-inducing factor (AIF) relocation in T lymphocytes entering the early commitment phase to apoptosis. J Biol Chem 278(33): 31401–31411 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

