Extrasynaptic delta-containing GABAA receptors in the nucleus accumbens dorsomedial shell contribute to alcohol intake
- PMID: 21368141
- PMCID: PMC3060220
- DOI: 10.1073/pnas.1016156108
Extrasynaptic delta-containing GABAA receptors in the nucleus accumbens dorsomedial shell contribute to alcohol intake
Abstract
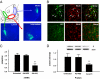
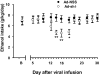
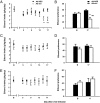
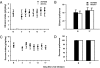
Recent findings suggest that extrasynaptic δ-subunit-containing GABA(A) receptors are sensitive to low-to-moderate concentrations of alcohol, raising the possibility that these receptors mediate the reinforcing effects of alcohol after consumption of one or a few drinks. We used the technique of viral-mediated RNAi to reduce expression of the GABA(A) receptor δ-subunit in adult rats in localized regions of the nucleus accumbens (NAc) to test the hypothesis that δ-subunit-containing GABA(A) receptors in the NAc are necessary for oral alcohol consumption. We found that knockdown of the δ-subunit in the medial shell region of the NAc, but not in the ventral or lateral shell or in the core, reduced alcohol intake. In contrast, δ-subunit knockdown in the medial shell did not affect intake of a 2% sucrose solution, suggesting that the effects of GABA(A) receptor δ-subunit reduction are specific to alcohol. These results provide strong evidence that extrasynaptic δ-subunit-containing GABA(A) receptors in the medial shell of the NAc are critical for the reinforcing effects of oral ethanol.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Comment in
-
Extrasynaptic GABAA receptors in the nucleus accumbens are necessary for alcohol drinking.Proc Natl Acad Sci U S A. 2011 Mar 22;108(12):4699-700. doi: 10.1073/pnas.1102818108. Epub 2011 Mar 9. Proc Natl Acad Sci U S A. 2011. PMID: 21389267 Free PMC article. No abstract available.
Similar articles
-
Activation of extrasynaptic δ-GABAA receptors globally or within the posterior-VTA has estrous-dependent effects on consumption of alcohol and estrous-independent effects on locomotion.Horm Behav. 2017 Sep;95:65-75. doi: 10.1016/j.yhbeh.2017.07.015. Epub 2017 Sep 20. Horm Behav. 2017. PMID: 28765080 Free PMC article.
-
Extrasynaptic GABAA receptors in the nucleus accumbens are necessary for alcohol drinking.Proc Natl Acad Sci U S A. 2011 Mar 22;108(12):4699-700. doi: 10.1073/pnas.1102818108. Epub 2011 Mar 9. Proc Natl Acad Sci U S A. 2011. PMID: 21389267 Free PMC article. No abstract available.
-
Alpha4-containing GABAA receptors in the nucleus accumbens mediate moderate intake of alcohol.J Neurosci. 2009 Jan 14;29(2):543-9. doi: 10.1523/JNEUROSCI.3199-08.2009. J Neurosci. 2009. PMID: 19144854 Free PMC article.
-
Alpha4 subunit-containing GABAA receptors in the accumbens shell contribute to the reinforcing effects of alcohol.Addict Biol. 2012 Mar;17(2):309-21. doi: 10.1111/j.1369-1600.2011.00333.x. Epub 2011 Apr 20. Addict Biol. 2012. PMID: 21507158 Free PMC article.
-
Effect of nucleus accumbens shell infusions of ganaxolone or gaboxadol on ethanol consumption in mice.Psychopharmacology (Berl). 2015 Apr;232(8):1415-26. doi: 10.1007/s00213-014-3777-x. Epub 2014 Oct 25. Psychopharmacology (Berl). 2015. PMID: 25342197 Free PMC article.
Cited by
-
Effect of ganaxolone and THIP on operant and limited-access ethanol self-administration.Neuropharmacology. 2012 Sep;63(4):555-64. doi: 10.1016/j.neuropharm.2012.05.007. Epub 2012 May 18. Neuropharmacology. 2012. PMID: 22613838 Free PMC article.
-
Nicotine decreases ethanol-induced dopamine signaling and increases self-administration via stress hormones.Neuron. 2013 Aug 7;79(3):530-40. doi: 10.1016/j.neuron.2013.06.006. Epub 2013 Jul 18. Neuron. 2013. PMID: 23871233 Free PMC article.
-
Activation of extrasynaptic δ-GABAA receptors globally or within the posterior-VTA has estrous-dependent effects on consumption of alcohol and estrous-independent effects on locomotion.Horm Behav. 2017 Sep;95:65-75. doi: 10.1016/j.yhbeh.2017.07.015. Epub 2017 Sep 20. Horm Behav. 2017. PMID: 28765080 Free PMC article.
-
Recent Perspectives on Sex Differences in Compulsion-Like and Binge Alcohol Drinking.Int J Mol Sci. 2021 Apr 6;22(7):3788. doi: 10.3390/ijms22073788. Int J Mol Sci. 2021. PMID: 33917517 Free PMC article. Review.
-
The incentive amplifying effects of nicotine: Roles in alcohol seeking and consumption.Adv Pharmacol. 2022;93:171-218. doi: 10.1016/bs.apha.2021.10.008. Epub 2022 Jan 11. Adv Pharmacol. 2022. PMID: 35341566 Free PMC article. Review.
References
-
- Grobin AC, Matthews DB, Devaud LL, Morrow AL. The role of GABAA receptors in the acute and chronic effects of ethanol. Psychopharmacology (Berl) 1998;139:2–19. - PubMed
-
- Chester JA, Cunningham CL. GABAA receptor modulation of the rewarding and aversive effects of ethanol. Alcohol. 2002;26:131–143. - PubMed
-
- Koob GF. A role for GABA mechanisms in the motivational effects of alcohol. Biochem Pharmacol. 2004;68:1515–1525. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

