Therapeutic targeting of SPINK1-positive prostate cancer
- PMID: 21368222
- PMCID: PMC3211047
- DOI: 10.1126/scitranslmed.3001498
Therapeutic targeting of SPINK1-positive prostate cancer
Abstract
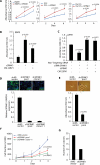
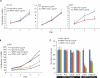
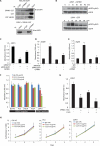
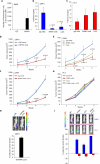
Gene fusions involving ETS (erythroblastosis virus E26 transformation-specific) family transcription factors are found in ~50% of prostate cancers and as such can be used as a basis for the molecular subclassification of prostate cancer. Previously, we showed that marked overexpression of SPINK1 (serine peptidase inhibitor, Kazal type 1), which encodes a secreted serine protease inhibitor, defines an aggressive molecular subtype of ETS fusion-negative prostate cancers (SPINK1+/ETS⁻, ~10% of all prostate cancers). Here, we examined the potential of SPINK1 as an extracellular therapeutic target in prostate cancer. Recombinant SPINK1 protein (rSPINK1) stimulated cell proliferation in benign RWPE as well as cancerous prostate cells. Indeed, RWPE cells treated with either rSPINK1 or conditioned medium from 22RV1 prostate cancer cells (SPINK1+/ETS⁻) significantly increased cell invasion and intravasation when compared with untreated cells. In contrast, knockdown of SPINK1 in 22RV1 cells inhibited cell proliferation, cell invasion, and tumor growth in xenograft assays. 22RV1 cell proliferation, invasion, and intravasation were attenuated by a monoclonal antibody (mAb) to SPINK1 as well. We also demonstrated that SPINK1 partially mediated its neoplastic effects through interaction with the epidermal growth factor receptor (EGFR). Administration of antibodies to SPINK1 or EGFR (cetuximab) in mice bearing 22RV1 xenografts attenuated tumor growth by more than 60 and 40%, respectively, or ~75% when combined, without affecting PC3 xenograft (SPINK1⁻/ETS⁻) growth. Thus, this study suggests that SPINK1 may be a therapeutic target in a subset of patients with SPINK1+/ETS⁻ prostate cancer. Our results provide a rationale for both the development of humanized mAbs to SPINK1 and evaluation of EGFR inhibition in SPINK1+/ETS⁻ prostate cancers.
Figures
Comment in
-
A two-step toward personalized therapies for prostate cancer.Sci Transl Med. 2011 Mar 2;3(72):72ps7. doi: 10.1126/scitranslmed.3002169. Sci Transl Med. 2011. PMID: 21368221 Free PMC article.
-
Role of the tumor-associated trypsin inhibitor SPINK1 in cancer development.Asian J Androl. 2011 Jul;13(4):628-9. doi: 10.1038/aja.2011.45. Epub 2011 May 23. Asian J Androl. 2011. PMID: 21602832 Free PMC article. No abstract available.
-
Words of wisdom. Re: Therapeutic targeting of SPINK1-positive prostate cancer.Eur Urol. 2012 Oct;62(4):733-4. doi: 10.1016/j.eururo.2012.07.021. Eur Urol. 2012. PMID: 22939424 No abstract available.
References
-
- Jemal A, Siegel R, Xu J, Ward E. Cancer statistics, 2010. CA Cancer J Clin. 2010;60:277–300. - PubMed
-
- Helgeson BE, Tomlins SA, Shah N, Laxman B, Cao Q, Prensner JR, Cao X, Singla N, Montie JE, Varambally S, Mehra R, Chinnaiyan AM. Characterization of TMPRSS2:ETV5 and SLC45A3:ETV5 gene fusions in prostate cancer. Cancer Res. 2008;68:73–80. - PubMed
-
- Tomlins SA, Rhodes DR, Perner S, Dhanasekaran SM, Mehra R, Sun XW, Varambally S, Cao X, Tchinda J, Kuefer R, Lee C, Montie JE, Shah RB, Pienta KJ, Rubin MA, Chinnaiyan AM. Recurrent fusion of TMPRSS2 and ETS transcription factor genes in prostate cancer. Science. 2005;310:644–648. - PubMed
-
- Tomlins SA, Mehra R, Rhodes DR, Smith LR, Roulston D, Helgeson BE, Cao X, Wei JT, Rubin MA, Shah RB, Chinnaiyan AM. TMPRSS2:ETV4 gene fusions define a third molecular subtype of prostate cancer. Cancer Res. 2006;66:3396–3400. - PubMed
-
- Tomlins SA, Laxman B, Dhanasekaran SM, Helgeson BE, Cao X, Morris DS, Menon A, Jing X, Cao Q, Han B, Yu J, Wang L, Montie JE, Rubin MA, Pienta KJ, Roulston D, Shah RB, Varambally S, Mehra R, Chinnaiyan AM. Distinct classes of chromosomal rearrangements create oncogenic ETS gene fusions in prostate cancer. Nature. 2007;448:595–599. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

