Functional analysis of CTRP3/cartducin in Meckel's cartilage and developing condylar cartilage in the fetal mouse mandible
- PMID: 21371032
- PMCID: PMC3089748
- DOI: 10.1111/j.1469-7580.2011.01354.x
Functional analysis of CTRP3/cartducin in Meckel's cartilage and developing condylar cartilage in the fetal mouse mandible
Abstract
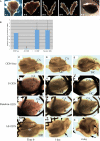
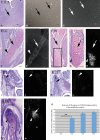
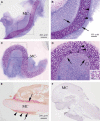
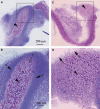
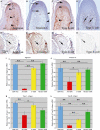
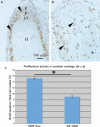
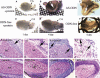
CTRP3/cartducin, a novel C1q family protein, is expressed in proliferating chondrocytes in the growth plate and has an important role in regulating the growth of both chondrogenic precursors and chondrocytes in vitro. We examined the expression of CTRP3/cartducin mRNA in Meckel's cartilage and in condylar cartilage of the fetal mouse mandible. Based on in situ hybridization studies, CTRP3/cartducin mRNA was not expressed in the anlagen of Meckel's cartilage at embryonic day (E)11.5, but it was strongly expressed in Meckel's cartilage at E14.0, and then reduced in the hypertrophic chondrocytes at E16.0. CTRP3/cartducin mRNA was not expressed in the condylar anlagen at E14.0, but was expressed in the upper part of newly formed condylar cartilage at E15.0. At E16.0, CTRP3/cartducin mRNA was expressed from the polymorphic cell zone to the upper part of the hypertrophic cell zone, but was reduced in the lower part of the hypertrophic cell zone. CTRP3/cartducin-antisense oligodeoxynucleotide (AS-ODN) treatment of Meckel's cartilage and condylar anlagen from E14.0 using an organ culture system indicated that, after 4-day culture, CTRP3/cartducin abrogation induced curvature deformation of Meckel's cartilage with loss of the perichondrium and new cartilage formation. Aggrecan, type I collagen, and tenascin-C were simultaneously immunostained in this newly formed cartilage, indicating possible transformation from the perichondrium into cartilage. Further, addition of recombinant mouse CTRP3/cartducin protein to the organ culture medium with AS-ODN tended to reverse the deformation. These results suggest a novel function for CTRP3/cartducin in maintaining the perichondrium. Moreover, AS-ODN induced a deformation of the shape, loss of the perichondrium/fibrous cell zone, and disorder of the distinct architecture of zones in the mandibular condylar cartilage. Additionally, AS-ODN-treated condylar cartilage showed reduced levels of mRNA expression of aggrecan, collagen types I and X, and reduced BrdU-incorporation. These results suggest that CTRP3/cartducin is not only involved in the proliferation and differentiation of chondrocytes, but also contributes to the regulation of mandibular condylar cartilage.
© 2011 The Authors. Journal of Anatomy © 2011 Anatomical Society of Great Britain and Ireland.
Figures


References
-
- Akiyama H, Furukawa S, Wakisaka S, et al. Cartducin stimulates mesenchymal chondroprogenitor cell proliferation through both extracellular signal-regulated kinase and phosphatidylinositol 3-kinase/Akt pathways. FEBS J. 2006;273:2257–2263. - PubMed
-
- Akiyama H, Furukawa S, Wakisaka S, et al. Elevated expression of CTRP3/cartducin contribute to promotion of osteosarcoma cell proliferation. Oncol Rep. 2009;21:1477–1481. - PubMed
-
- Beresford WA. Secondary cartilages I, II. In: Beresford WA, editor. Chondroid Bone, Secondary Cartilage and Metaplasia. Baltimore: Urban and Schwarzenberg; 1981. pp. 23–65.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

